While there are mechanisms controlling transcription of DNA as discussed above, there are also mechanisms, which operate after the transcription is initiated. For instance, in
tryptophan operon of
E. coli, the mRNA of gene
trp E (gene for enzyme anthranilate synthetase) has a sequence of 162 bases between its initiation codon AUG and the promoter-operator region of this gene. This sequence of 162 bases is called
'leader' sequence and has several features of interest, which determine post-transcriptional control. (This post-transcriptional control is in addition to the normal transcriptional control provided in operon model). The leader sequence at its 3' end, but 8-12 bases before the initiating codon, has a sequence 5'AGGAGGA 3',which is common to several
E. coli genes and was discovered by J.
Shine and L.
Delgarno. This sequence 5'AGGAGGA 3' (known as
Shine Delgarno sequence) base pairs with the sequence 5'-ACC'UCC'UUA-OH-3' found at 3' end of 16S rRNA and thus helps in mRNA binding to ribosome.
Besides the sequence 5'AGGAGGA 3' for ribosome binding, there are other sequences of interest in the leader sequence. In leader sequence of
trp E discussed above, a mechanism is available, which allows further transcription when synthesis is needed, and stops the transcription whenever the synthesis is not needed. For instance, when tryptophan is in short supply, so that its further supply is needed, the transcription will take place and when tryptophan is in adequate supply, so that its further supply is not needed, the transcription will stop. This is achieved by a unique feature common to several operons meant for synthesis of amino acids.
This unique feature includes successive tryptophan codons at a specific site in
trp E leader sequence. Similarly, in leader sequence of
phenylalanine operon, there are three consecutive phenylalanine codons; in leader sequence of
leucine operon, there are four consecutive leucine codons and in leader sequence
of histidine operon, there are eight consecutive histidine codons. These regions of leader sequences each having consecutive codons for the same amino acid, for the synthesis of which the operon is meant, gives rise to mRNA for a small polypeptide about 15 amino acid residues long. Significance of-.this polypeptide synthesized by leader sequence has been explained as follows.
In tryptophan operon, it has been shown that when tryptophan supply is adequate, transcription stops at about 147 bases after initiation, so that no mRNA for
trp E and other genes of the operon are formed. But if tryptophan is in short supply, transcription continues uninhibited. During adequate supply of tryptophan, mRNA for leader sequence is translated on ribosomes without any difficulty since two consecutive tryptophan codons mentioned above will be easily read. This favourable synthesis of leader peptide leads to the formation of hairpin loop, also known as
attenuator, in the sequence between bases 114 and 134 (Figs. 35.22 and 35.23). This stops further synthesis of mRNA. On the other hand, if tryptophan is in short supply, the two
trp codons (UGG, UGG) can not be translated due to lack of availability of amino acid
'trp' and thus ribosome becomes stalled at this point (Fig. 35.24) and makes
the formation of
terminator hairpin loop (in sequence 114-134) impossible, thus indirectly permitting transcription of
trpE and other genes (Fig. 35.25).
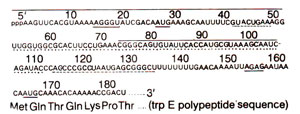
Fig. 35.22. The leader sequence of mRNA transcribed from E. coli trp (tryptophan)operon. Specific regions for signals are underlined. Probable ribosomes binding Shine-Delgarno sequences at 15-19 and 152-155 nucleotides; at 27-29 and 165-167, there is initiating codon AUG; two adjoining trp codons (UGG UGG) at 54-59; sequence 114-134, underlined by broken line, is termination structure (attenuator) shown in Fig. 35.23.
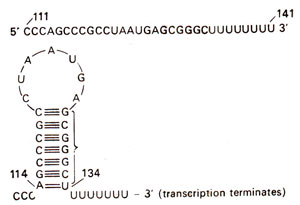
Fig. 35.23. Termination structure (stem and loop), which is shown by broken underline in Fig. 35.22

Fig. 35.24. Different alternatives for RNA polymerase due to position of ribosome, which depends on hairpin formation.
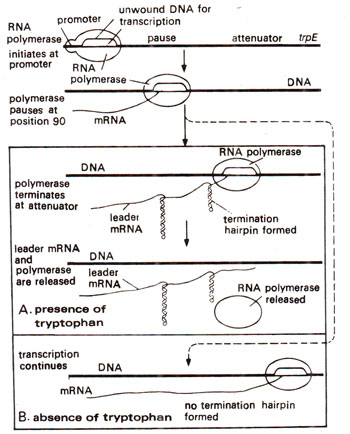
Fig. 35.25. Role of attenuator in regulation of the transcription on tryptophan operon. (A) The presence of tryptophan allows formation of hairpin leading to release of RNA polymerase. (B) The absence of tryptophan allows the polymerase to continue transcription (for details see text).
The leader sequences, thus seem to have a function of attenuating (reducing) the transcription in a controlled way. It should, however be realized that such a transcriptional regulation is not possible in eukaryotes since it depends on coupling of translation with transcription and in eukaryotes, translation and transcription can not be coupled due to separation by nuclear envelope.








