N-Linked Glycoproteins
N-linked glycoproteins have carbohydrate covalently attached to asparagine residues that occur in the sequence Asn-X-Ser/Thr, where X is any residue except proline. This is a necessary but not sufficient key for glycosylation since there are many examples of such sequences that are not glycosylated even when others on the same polypeptide are substituted with sugar. The linking sugar is invariably N-acetylglucosamine, which is the terminal saccharide of the attached unit (Fig. 25). The number of saccharides present in N-linked structures varies from about 7 to 20 or more; branching is universal with some structures having four separate branches (antennae). All of the saccharides have a common core structure: GlcNAc- GlcNAc-Man3. The first mannose is β-linked (unusual) and the other two mannoses are attached α-1-3 and α-1-6, thus forming the initial branch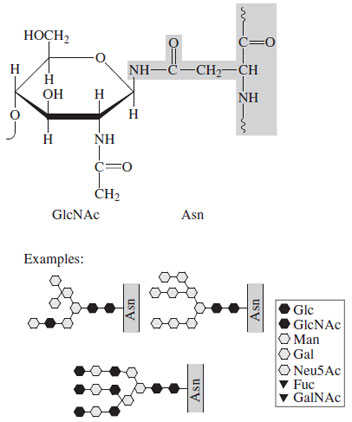 |
| Figure 25 Schematic of a typical N-linked oligosaccharide. Note the core structure, which contains two N-acetylglucosamine and three mannosyl residues. This is present in all units of this type. |
The biosynthesis of these molecules is also unusual. The saccharide is preassembled, not as the final structure but as a common, 14-sugar, lipiD-linked precursor that is transferred en bloc to the target asparagine in a cotranslational manner (Fig. 26). This saccharide unit (GlcNAc2-Man9- Glc3) is trimmed to a GlcNAc2-Man5 structure that is then modified by addition of either more mannosyl residues or by several sugars, including GlcNAc, Gal, NANA, and Lfucose. The latter category is generally termed complex as opposed to those which contain GlcNAc and Man only (high mannose).
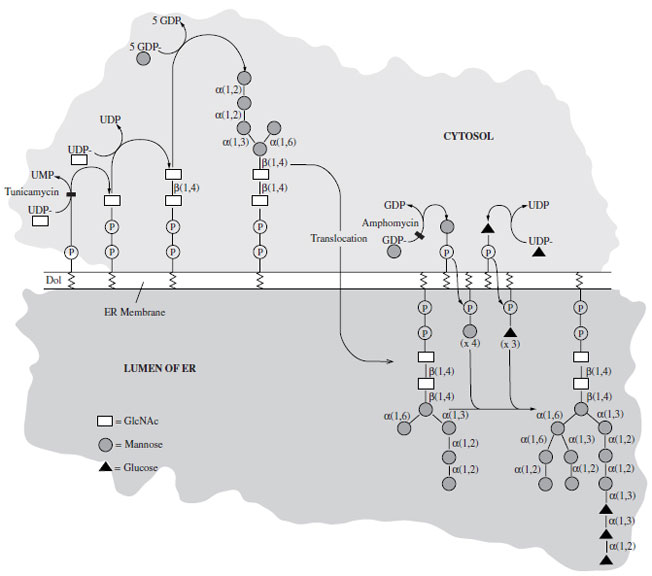 |
| Figure 26 Biosynthesis of the 14-sugar, lipiD-linked oligosaccharide, the universal precursor for N-linked glycosylation. |




