Primary Culture of Drosophila Embryo Cells
I. INTRODUCTION |
| FIGURE 1 Large plastic jars used for embryo collections. The jar on the left is empty and the one on the right has a grape juice agar embryo collection plate taped to the bottom and is stocked with about 3000 adult flies as described. Holes have been cut into the sides of the jars and are covered with nylon mesh to allow efficient air circulation. |
Primary cultures of Drosophila embryonic cells offer a unique system to study the transitions of undifferentiated cells into a variety of cell types. Coupled with the power of Drosophila classical and molecular genetics, the analyses that are possible in vitro using differentiating embryo cells will continue to contribute to a deeper understanding of development. Preparation of primary embryo cell cultures was developed independently in the laboratories of Seecof and colleagues (Seecof and Unanue, 1968) and Shields and Sang (1970). The procedures for preparing cultures are technically undemanding and can be adapted to solve a wide range of biological problems. Important variations include the culture of single embryos (Cross and Sang, 1978), as well as the development of techniques for isolating the precursors of different cell types such as neurons and muscle cells (Furst and Mahowald, 1985; Hayashi and Perez-Magallanes, 1994). Figure 1 shows a convenient way to collect embryos from large populations of egglaying adults, and Fig. 2 illustrates a typical setup used for preparing primary embryo cultures.
 |
| FIGURE 2 Typical items used to prepare Drosophila gastrula stage embryo cultures are arranged in a small laminar flow hood. |
Penicillin and streptomycin are from GIBCO/BRL (Cat. No. 15140-122). Insulin is from Sigma (Cat. No. 1- 5500). 1X Schneider's Drosophila medium with glutamine is from GIBCO (Cat. No. 11720-034). Fetal bovine serum is from GIBCO (Cat. No. 10082-139).
Cells are usually cultured in 35-mm plastic dishes from Corning (Cat. No. 430165). For finer morphological observations, cells are plated directly onto round glass coverslips placed in the bottom of a 35-mm plate (five per culture dish, Fisher Scientific, Cat. No. 12-545- 801-1THK). Coverslips are presterilized using standard gas or autoclave procedures.
Dechorionated embryos are dissociated in 15-ml Dounce glass-glass homogenizers from Baxter Diagnostics (Cat. No. T4018-15). Nylon mesh (25-100 µm pour size) can be obtained from most fabric stores. All other equipment and reagents are commonly used for general laboratory work or tissue culture and can be obtained from any scientific supply company.
A. Egg Collection and Aging to Early Gastrula Stage
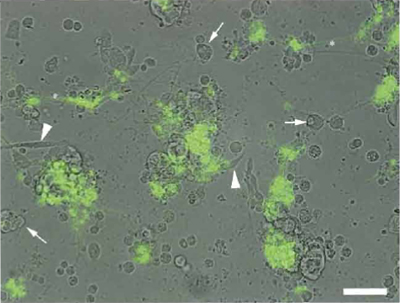 |
| FIGURE 3 A Drosophila gastrula stage embryo cell culture after about 2 days of in vitro differentiation. The majority of cells differentiate into neurons organized in clusters. This image is a composite of a bright-field image overlaid with a false-colored fluorescent image of the same field. Green cells are neurons expressing a cholinergic-specific fluorescent transgene and are indicative of cells with a cholinergic neurotransmitter phenotype. White arrowheads show some multinuicleate myotubes, white and the asterisk indicates axonallike processes emanating from neurons or neural clusters and are often connected to neurons in adjacent clusters. Scale bar: 50 µm. |
Yeast-proprionic acid paste: Dissolve dry yeast in 2% proprionic acid
Grape-juice-agar egg collection plates: 160ml water, 160ml grape juice (we use Welch's), 6.4 g Bacto-agar (Becton, Dickinson, Cat. No. 214010), 0.8g Nipagin [p-hydroxybenzoic acid methyl ester (methylpraben)] (Sigma, Cat. No. H-5501). Combine water, grape juice, and Bacto-agar in a l-liter flask. Microwave to dissolve agar, being careful not to let the solution boil over. Remove from the microwave, add Nipagin, mix, and pour into 12 large (14cm) culture plates.
Steps
- Prepare large egg-laying population by transferring 1.5-week-old adult flies from 8 to 12 ordinary culture bottles to fresh culture bottles supplemented with dry yeast sprinkled over the food. Allow the population to adapt for 1-2 days in a 25°C humidified room and transfer the adults to an embryo collection bottle on the day cultures will be prepared. Figure 1 shows an example of a homemade collection bottle or a similar one can be obtained commercially (Doc Frugal Scientific, Cat. No. 55-100). We generally stock each egg-laying population with about 3000 adult flies. For convenience, maintain the room on a reverse light-dark cycle so collections can be done in the investigators' morning (i.e., the flies' dusk).
- Apply a small amount of yeast-proprionic acid paste on a small piece of filter paper and place in the center of a large (14-cm) grape juice-agar egg collection plate to attract females to lay eggs.
- Invert the embryo collection bottles onto the food plate and collect eggs for 1 or 2h.
- If better developmental timing (synchronization) is desired, do a 30-min to 1-h precollection and discard plates. This may be necessary to induce females to lay eggs they have been storing for longer times.
- Remove egg collection plates from the embryo collection bottles, cover, and allow embryos to develop to early gastrula stage at 25°C for 3.5 h.
Solution
Deionized water: Prepare 2 to 3 liters of deionized water.
Steps
- Embryos should be harvested 5-10min before the 3.5-h aging period so the cultures can be initiated immediately at 3.5 h.
- Discard filter papers with yeast mixture.
- Transfer plates to sink and rinse extensively and gently with deionized water to remove adult flies and other debris.
- Pour water on the plates and gently loosen embryos with a camels' hair paint brush.
- Pour suspended embryos into a nylon mesh filter to allow further washing and collection from other food plates.
- Continue washing and remove large food particles and other debris with forceps if necessary.
- Transfer nylon mesh with embryos to a sterile tissue culture hood.
C. Preparation of Embryonic Cultures
Penicillin-streptomycin stock solution: Penicillin-streptomycin solution (10,000U/ml) is obtained from GIBCO/BRL (Cat. No. 15140-122). Use 10ml per liter of medium (i.e., final concentration of 100 U / ml penicillin, 100 µg/ml streptomycin).
Insulin stock solution: Dissolve insulin in 0.05 N HCl at a final concentration of 2mg/ml. Filter sterilize (0.2 µm) and store at 4°C (stock is stable for 1 year). Use at a final concentration of 0.2 µg/ml of medium.
Schneider's Drosophila medium with glutamine, insulin, and 5% fetal bovine serum: Add 10ml of sterile penicillin/ streptomycin stock solution, 0.1 ml of insulin stock solution, and 50 ml of fetal bovine serum (final serum concentration = 5%) to 1 liter of Schneider's Drosophila medium with glutamine. The medium supports good differentiation when stored at 4°C for up to 1 month.
Sodium hypochlorite-ethanol: In a small beaker mix 10 ml of 95% EtOH and 10ml of 3% sodium hypochlorite. This solution will dechorionate the embryos and sterilize them.
Deionized water: Prepare about 1 liter of sterile deionized water.
- Preclean the laminar flow hood with ethanol using normal tissue culture procedures. All further operations are carried out under sterile conditions in the hood.
- Transfer embryos to the beaker of sodium hypochlorite-ethanol using a sterile spatula and swirl gently for 1 to 2min. The time may vary slightly but do not exceed 3 min. Timing depends on the freshness of the hypochlorite and the compromises you are willing to make with respect to yield and sterility. Shorter times tend toward better yields and longer times are necessary if you wish to totally eliminate yeast contamination.
- Pour dechorionated embryos onto a small nylon mesh filter funnel and wash extensively with about 500ml sterile distilled water.
- Transfer to a 15-ml Dounce homogenizer that contains about 6 ml of culture medium and dissociate cells by gently homogenizing with three rotary up and down strokes.
- Filter dissociated cells through a 25- to 100-µm nylon mesh filter (the mesh is taped loosely over a 5- cm-diameter glass funnel or fitted into a 3.5-cm metal ring that is placed into the funnel) to remove undissociated clumps and other large debris.
- Collect filtrate in a 15-ml centrifuge tube and pellet cells by centrifugation at 1400rpm for 4min. Wash cells one time in 6 ml of culture medium and centrifuge as described earlier, followed by a final suspension in 10 ml of culture medium.
- Place a small aliquot in a hemocytometer and count the round, intact, medium-sized cells and cell clusters to determine cell number.
- Calculate the concentration of cells, add sufficient medium to obtain a final density of 8 × 105 cells/ml, and plate approximately 1.6 × 106 cells in a 35-mm culture dish (i.e., 2ml of cell suspension per dish).
- Culture cells in a humidified 25°C incubator (CO2 is not necessary). Cells begin to differentiate in a few hours and are substantially developed after about 24h. Cells can be cultured for several days before replacing the medium. It is possible to keep cultures for more than 1 year.
Cells are usually grown in 35-mm dishes but it is also possible to plate directly onto coverslips (see earlier) and even to grow cells in the raised condensation rings found on the lids of many 96-well microtiter plates. This is especially convenient when screening monoclonal antibodies, as each embryo culture can be isolated and matched with the corresponding position for a hybridoma clone growing in a 96-well culture dish. A proceedure has been described for treating the lids of culture plates with H2SO4 that results in a significant improvement in the attachment and differentiation of Drosophila embryo cells (Furst and Mahowald, 1984).
The yield of cells depends on how vigorously the cells are dissociated and how long the embryos are exposed to the ethanol-hypochlorite mixture. Using the previously described procedures, we can routinely obtain ~106 cells for plating from a single embryo collection bottle stocked with about 3000 adult flies. This is sufficient for preparing five 35-mm culture dishes. The procedure can be scaled up or down easily using additional embryo collection bottles or using a smaller egg-laying population.
In the past we have used up to 20% fetal calf serum; however, such high concentrations are not necessary to support optimal growth and differentiation. It is especially important to use prescreened lots of fetal calf serum that are suitable for Drosophila embryo cultures. Some lots of fetal calf serum do not support differentiation and even appear to be toxic to Drosophila embryonic cells. We have had good success with serum from GIBCO and Gemini Products. A chemically defined medium for preparing embryo cultures has also been described (O'Dowd, 1995). The surface coating of tissue culture plasticware varies from different manufacturers. We have found that some types of 35-mm plastic dishes, such as those from Falcon, are not optimal for Drosophila cultures.
Acknowledgments
Work in the authors' laboratories has been supported by grants from NIH-NINDS. We thank our past and current laboratory colleagues for many helpful suggestions and ideas about Drosophila primary embryo cultures, especially Drs. N. Bournias- Vardiabasis and S. Song.
Brody, T., and Odenwald, W. E (2000). Programmed transformations in neuroblast gene expression during Drosophila CNS lineage development. Dev. Biol. 226, 34-44.
Brody, T., and Odenwald, W. E (2002). Cellular diversity in the developing nervous system: A temporal view from Drosophila. Development 129, 3763-3770.
Byerly, L., and Leung, H. T. (1988). Ionic currents of Drosophila neurons in embryonic cultures. J. Neurosci. 8, 4379-4393.
Cross, D. P., and Sang, J. H. (1978). Cell culture of individual Drosophila embryos. I. Development of wild-type cultures. J. Embryol. Exp. Morphol. 45, 161-172.
Furst, A., and Mahowald, A. P. (1984). Rapid immunofluorescent screening procedure using primary cell cultures or tissue sections. J. Immunol. Methods 70, 101-109.
Furst, A., and Mahowald, A. P. (1985). Differentiation of primary embryonic neuroblasts in purified neural cell cultures from Drosophila. Dev. Biol. 109, 184-192.
Hayashi, I., and Perez-Magallanes, M. (1994). Establishment of pure neuronal and muscle precursor cell cultures from Drosophila early gastrula stage embryos. In Vitro Cell Dev. Biol. Anim. 30A, 202-208.
Hayashi, I., Perez-Magallanes, M., and Rossi, J. M. (1992). Neurotrophic factor-like activity in Drosophila. Biochem. Biophys. Res. Commun. 184, 73-79.
Lee, D., and O'Dowd, D. K. (1999). Fast excitatory synaptic transmission mediated by nicotinic acetylcholine receptors in Drosophila neurons. J. Neurosci. 19, 5311-5321.
O'Dowd, D. K. (1995). Voltage-gated currents and firing properties of embryonic Drosophila neurons grown in a chemically defined medium. J. Neurobiol. 27, 113-126.
Salvaterra, P. M., Bournias-Vardiabasis, N., Nair, T., Hou, G., and Lieu, C. (1987). In vitro neuronal differentiation of Drosophila embryo cells. J. Neurosci. 7, 10-22.
Seecof, R. L., Gerson, I., Donady, J. J., and Teplitz, R. L. (1973b). Drosophilia myogenesis in vitro: The genesis of "small" myocytes and myotubes. Dev. Biol. 35, 250-261.
Seecof, R. L., Teplitz, R. L., Gerson, I., Ikeda, K., and Donady, J. (1972). Differentiation of neuromuscular junctions in cultures of embryonic Drosophila cells. Proc. Natl. Acad. Sci. USA 69, 566-570.
Seecof, R. L., and Unanue, R. L. (1968). Differentiation of embryonic Drosophila cells in vitro. Exp. Cell Res. 50, 654-660.
Shields, G., Dubendorfer, A., and Sang, J. H. (1975). Differentiation in vitro of larval cell types from early embryonic cells of Drosophila melanogaster. J. Embryol. Exp. Morphol. 33, 159-175.
Shields, G., and Sang, J. H. (1970). Characteristics of five cell types appearing during in vitro culture of embryonic material from Drosophila melanogaster. J. Embryol. Exp. Morphol. 23, 53-69.




