Invasion of Fresh Water
Invasion of Fresh Water
Some 400 million years ago, during the Silurian and Lower Devonian periods, the major groups of jawed fishes began to penetrate into brackish-water estuaries and then gradually into freshwater rivers. Before them lay a new, unexploited habitat already stocked with food in the form of insects and other invertebrates, which had preceded them into fresh water. However, the advantages of this new habitat were balanced by a tough physiological challenge: the necessity of developing effective osmotic regulation.
Freshwater animals must keep the salt concentration of their body fluids higher than that of the water in which they live. Water enters their bodies osmotically, and salt is lost by diffusionoutward. Their problems are similar to those of the brackish-water crab, but more severe and unremitting. Fresh water is much more dilute than are coastal estuaries, and there is no retreat, no salty sanctuary into which a freshwater animal can retire for osmotic relief. It must and has become a permanent and highly efficient hyperosmotic regulator.
The scaled and mucus-covered body surface of a fish is about as waterproof as any flexible surface can be. In addition, freshwater fishes have several defenses against the problems of water gain and salt loss. First, water that inevitably enters by osmosis across the gills is pumped out by the kidney, which is capable of forming very dilute urine (Figure 32-3). Second, special salt-absorbing cells located in the gills move salt ions, principally sodium and chloride (present in small quantities even in fresh water), from the water to the blood. This process, together with salt present in the fish’s food, replaces diffusive salt loss. These mechanisms are so efficient that a freshwater fish devotes only a small part of its total energy expenditure to maintain osmotic balance.
Crayfishes, aquatic insect larvae,
clams, and other freshwater animals
are also hyperosmotic regulators and
face the same hazards as freshwater
fishes; they tend to gain too much
water and lose too much salt. Like
freshwater fishes, they solve these
problems by excreting excess water as
urine and replacing lost salt by some
salt-transporting mechanism on the
body surface.
Amphibians living in water also must compensate for salt loss by actively absorbing salt from the water (Figure 32-4). They use their skin for this purpose. Physiologists learned some years ago that pieces of frog skin continue to
transport sodium and chloride actively for hours when removed and placed in a specially balanced salt solution. Fortunately for biologists, but unfortunately for frogs, these animals are so easily collected and maintained in the laboratory that frog skin became a favorite membrane system for studies of ion-transport phenomena.
Some 400 million years ago, during the Silurian and Lower Devonian periods, the major groups of jawed fishes began to penetrate into brackish-water estuaries and then gradually into freshwater rivers. Before them lay a new, unexploited habitat already stocked with food in the form of insects and other invertebrates, which had preceded them into fresh water. However, the advantages of this new habitat were balanced by a tough physiological challenge: the necessity of developing effective osmotic regulation.
Freshwater animals must keep the salt concentration of their body fluids higher than that of the water in which they live. Water enters their bodies osmotically, and salt is lost by diffusionoutward. Their problems are similar to those of the brackish-water crab, but more severe and unremitting. Fresh water is much more dilute than are coastal estuaries, and there is no retreat, no salty sanctuary into which a freshwater animal can retire for osmotic relief. It must and has become a permanent and highly efficient hyperosmotic regulator.
The scaled and mucus-covered body surface of a fish is about as waterproof as any flexible surface can be. In addition, freshwater fishes have several defenses against the problems of water gain and salt loss. First, water that inevitably enters by osmosis across the gills is pumped out by the kidney, which is capable of forming very dilute urine (Figure 32-3). Second, special salt-absorbing cells located in the gills move salt ions, principally sodium and chloride (present in small quantities even in fresh water), from the water to the blood. This process, together with salt present in the fish’s food, replaces diffusive salt loss. These mechanisms are so efficient that a freshwater fish devotes only a small part of its total energy expenditure to maintain osmotic balance.
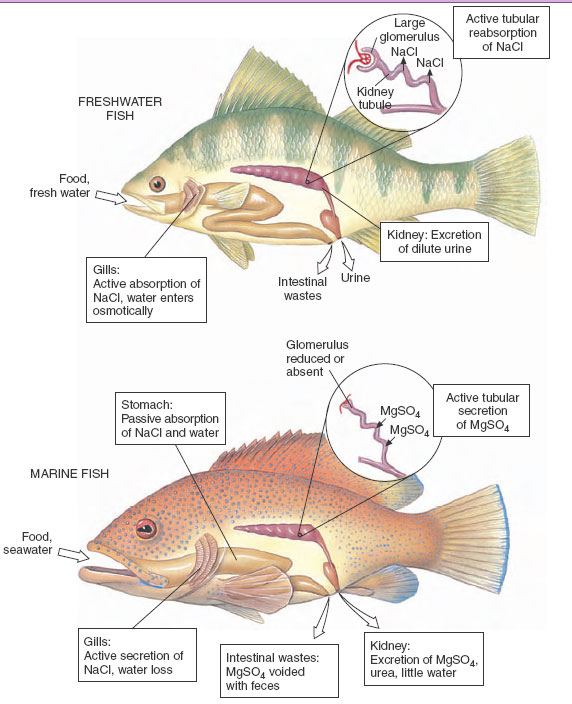 |
| Figure 32-3 Osmotic regulation in freshwater and marine bony fishes. A freshwater fish maintains osmotic and ionic balance in its dilute environment by actively absorbing sodium chloride across the gills (some salt enters with food). To flush out excess water that constantly enters the body, the glomerular kidney produces a dilute urine by reabsorbing sodium chloride. A marine fish must drink seawater to replace water lost osmotically to its salty environment. Sodium chloride and water are absorbed from the stomach. Excess sodium chloride is secreted outward by the gills. Divalent sea salts, mostly magnesium sulfate, are eliminated with feces and secreted by the tubular kidney. |
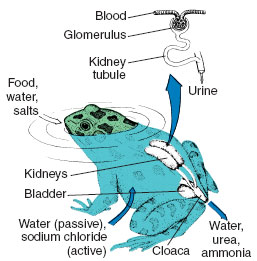 |
| Figure 32-4 Exchange of water and solute in a frog. Water enters the highly permeable skin and is excreted by the kidney. The skin also actively transports ions (sodium chloride) from the environment. The kidney forms a dilute urine by reabsorbing sodium chloride. Urine flows into the urinary bladder , where, during temporary storage, most of the remaining sodium chloride is removed and returned to the blood. |
Amphibians living in water also must compensate for salt loss by actively absorbing salt from the water (Figure 32-4). They use their skin for this purpose. Physiologists learned some years ago that pieces of frog skin continue to
transport sodium and chloride actively for hours when removed and placed in a specially balanced salt solution. Fortunately for biologists, but unfortunately for frogs, these animals are so easily collected and maintained in the laboratory that frog skin became a favorite membrane system for studies of ion-transport phenomena.




