Water and Osmotic Regulation
Water and Osmotic
Regulation
How Marine Invertebrates Meet Problems of Salt and Water Balance
Most marine invertebrates are in osmotic equilibrium with their seawater environment. They have body surfaces that are permeable to salts and water so that their body fluid concentration rises or falls in conformity with changes in concentrations of seawater. Because such animals are incapable of regulating osmotic pressure of their body fluid, they are called osmotic conformers. Invertebrates living in the open sea are seldom exposed to osmotic fluctuations because the ocean is a highly stable environment. Oceanic invertebrates have, in fact, very limited abilities to withstand osmotic change. If they should be exposed to dilute seawater, they die quickly because their body’s cells cannot tolerate dilution and are helpless to prevent it. These animals are restricted to living in a narrow salinity range and are said to be stenohaline (Gr. stenos, narrow, + hals, salt). An example is the marine spider crab (Figure 32-2).
Conditions along coasts and in estuaries and river mouths are much less constant than those of the open ocean. Here animals must be able to withstand large and often abrupt changes in salinity as the tides ebb and flow and mix with fresh water draining from rivers. These animals are termed euryhaline (Gr. eurys, broad, + hals, salt), meaning that they can survive a wide range of salinity changes, mainly because they demonstrate varying powers of osmotic regulation. For example, the brackishwater shore crab can resist dilution of body fluids by dilute (brackish) seawater (Figure 32-2). Although the concentration of salts in the body fluids falls, it does so less rapidly than the fall in seawater concentration. This crab is a hyperosmotic regulator, meaning that it maintains its body fluids more concentrated (hence hyper-) than the surrounding water.
By regulating against excessive dilution, thus protecting the cells from extreme changes, these crabs can live successfully in the physically unstable but biologically rich coastal environment. Nevertheless, with limited capacity for osmotic regulation, they will die if exposed to greatly diluted seawater. To understand how the brackish-water shore crab and other coastal invertebrates achieve hyperosmotic regulation, let us examine the problems they face. First, because the crab’s body fluids are osmotically more concentrated than the dilute seawater outside, water flows into its body, especially across the thin, permeable membranes of the gills. As with the membrane osmometer containing a salt solution , water diffuses inward because it is more concentrated outside than inside. For the crab, were this inflow of water allowed to continue unchecked, its body fluids would soon become diluted and unbalanced. The problem is solved by the kidneys (antennal glands located in the crab’s head), which can excrete the excess water as a dilute urine.
The second problem is salt loss. Again, because the animal is saltier than its environment, it cannot avoid loss of ions by outward diffusion across the gills. Salt is also lost in urine. This problem is solved by special saltsecreting cells in the gills that actively remove ions from dilute seawater and move them into the blood, thus maintaining the internal osmotic concentration. This is an active transport process that requires energy because ions must be transported against a concentration gradient from a lower salt concentration (in dilute seawater) to an already higher one (in blood).
How Marine Invertebrates Meet Problems of Salt and Water Balance
Most marine invertebrates are in osmotic equilibrium with their seawater environment. They have body surfaces that are permeable to salts and water so that their body fluid concentration rises or falls in conformity with changes in concentrations of seawater. Because such animals are incapable of regulating osmotic pressure of their body fluid, they are called osmotic conformers. Invertebrates living in the open sea are seldom exposed to osmotic fluctuations because the ocean is a highly stable environment. Oceanic invertebrates have, in fact, very limited abilities to withstand osmotic change. If they should be exposed to dilute seawater, they die quickly because their body’s cells cannot tolerate dilution and are helpless to prevent it. These animals are restricted to living in a narrow salinity range and are said to be stenohaline (Gr. stenos, narrow, + hals, salt). An example is the marine spider crab (Figure 32-2).
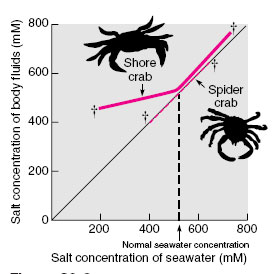 |
| Figure 32-2 Salt concentration of body fluids of two crabs as affected by variations in seawater concentration. The 45-degree line represents equal concentration between body fluids and seawater. Since the spider crab cannot regulate the salt concentration of its fluids, it conforms to whatever changes happen in the seawater. The shore crab, however , can regulate osmotic concentration of its fluids to some degree in dilute seawater. For example, when seawater is 200 mM (millimolar), the shore crab’s body fluid is approximately 430 mM. Crosses at ends of lines indicate limits of tolerance for each species. |
Conditions along coasts and in estuaries and river mouths are much less constant than those of the open ocean. Here animals must be able to withstand large and often abrupt changes in salinity as the tides ebb and flow and mix with fresh water draining from rivers. These animals are termed euryhaline (Gr. eurys, broad, + hals, salt), meaning that they can survive a wide range of salinity changes, mainly because they demonstrate varying powers of osmotic regulation. For example, the brackishwater shore crab can resist dilution of body fluids by dilute (brackish) seawater (Figure 32-2). Although the concentration of salts in the body fluids falls, it does so less rapidly than the fall in seawater concentration. This crab is a hyperosmotic regulator, meaning that it maintains its body fluids more concentrated (hence hyper-) than the surrounding water.
By regulating against excessive dilution, thus protecting the cells from extreme changes, these crabs can live successfully in the physically unstable but biologically rich coastal environment. Nevertheless, with limited capacity for osmotic regulation, they will die if exposed to greatly diluted seawater. To understand how the brackish-water shore crab and other coastal invertebrates achieve hyperosmotic regulation, let us examine the problems they face. First, because the crab’s body fluids are osmotically more concentrated than the dilute seawater outside, water flows into its body, especially across the thin, permeable membranes of the gills. As with the membrane osmometer containing a salt solution , water diffuses inward because it is more concentrated outside than inside. For the crab, were this inflow of water allowed to continue unchecked, its body fluids would soon become diluted and unbalanced. The problem is solved by the kidneys (antennal glands located in the crab’s head), which can excrete the excess water as a dilute urine.
The second problem is salt loss. Again, because the animal is saltier than its environment, it cannot avoid loss of ions by outward diffusion across the gills. Salt is also lost in urine. This problem is solved by special saltsecreting cells in the gills that actively remove ions from dilute seawater and move them into the blood, thus maintaining the internal osmotic concentration. This is an active transport process that requires energy because ions must be transported against a concentration gradient from a lower salt concentration (in dilute seawater) to an already higher one (in blood).




