Cell Cycles
The onion root tip and the whitefish blastula remain the standard introduction to the study of mitosis. The onion has easily observable chromosomes, and the whitefish has one of the clearest views of the spindle apparatus. The testis of the grasshopper and the developing zygote of the roundworm Ascaris are the traditional materials used for viewing the various stages of meiosis. In a single longitudinal section of a grasshopper testis, one can usually find all of the stages of meiotic development. The stages are also aligned from one pole of the testis. Few other meiotic samples are as convenient. For most materials, meiosis occurs in a more randomly distributed pattern throughout the testis.Ascaris is utilized to observe the final stages of development in eggs (oogenesis). The Ascaris egg lies dormant until fertilized. It then completes meiosis, forming 2 polar bodies while the sperm nucleus awaits fusion with the female nucleus. When this phenomenon is coupled with the large abundance of eggs in the Ascaris body, it makes an ideal specimen for observing the events of fertilization, polar body formation, fusion of pronuclei, and the subsequent division of the cell (cytokinesis).
Interphase G1-S-G2
The stages of mitosis were originally detailed after careful analysis of fixed cells. More recently, time-lapse photography coupled with phase-contrast microscopy has allowed us to visualize the process in its entirety, revealing a dynamic state of flux.
In early work, so much emphasis was placed on the movement of the chromosomes that the cell was considered to be “at rest” when not in mitosis. As significant as mitotic division is, it represents only a small fraction of the life span of a cell. Nonetheless, you may still come across the term “resting phase” in some older texts. This term is rarely used today, and the term interphase is sufficient for all activities between 2 mitotic divisions. The cell is highly active during interphase and most of the metabolic and genetic functions of the cell are reduced during the physical division of the nuclear and cellular materials (mitosis).
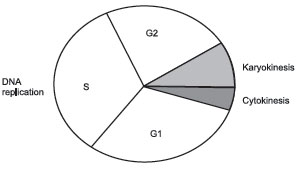
Interphase is divided into 3 subphases, G1, S, and G2. The basis for this division is the synthesis of DNA. While the entire cycle may be as long as 24 hours, mitosis is normally less than 1 hour in length. Because of the synthesis of DNA in interphase, the amount of DNA per nucleus is different depending on which subphase of interphase the cell is in. DNA can be measured using the fuelgen reaction and a microspectrophotometer. The basic amount of DNA in a haploid nucleus is given the value C. A diploid nucleus would be 2C. A triploid and tetraploid cell would be 3C and 4C, respectively.
However, when nuclei are actually measured, diploid cells in interphase can be divided into 3 groups; some are 2C and some are 4C, while a few are at intermediate values between 2C and 4C. The conclusion is that the genetic material (DNA) and presumably the chromosomes must duplicate. The period within interphase and during which DNA is synthesized is termed the S phase (for synthesis). The period of interphase preceding the S phase is the G1 phase (for 1st-Growth Phase), while the period subsequent to the S phase is the G2 phase (for 2nd-Growth Phase). During the G1 period, the cell is generally increasing in size and protein content. During S, the cell replicates the chromosomes and synthesizes DNA. During G2, it continues to increase in size, but also begins to build a significant pool of ATP and other high-energy phosphates, which are believed to be a significant part of the triggering mechanism for the subsequent karyokinetic and cytokinetic events of mitosis.
Mitosis returns the cells to the 2C state. Meiosis reduces the amount of DNA even further, to 1C. Meanwhile, the number of chromosomes (designated with the letter N) is also changing. For a diploid cell, the number of chromosomes is twice that of a haploid, or 2N. During mitosis, a diploid cell would go from one 2N cell to two 2N cells. Since the daughter cells have the same chromosome number as the parent, mitosis is also referred to as equational division. If a diploid (2N) cell undergoes meiosis, it will result in 4 haploid cells, each 1N.
Thus, meiosis is also referred to as reductional division. Refer to the figure below for comparison of C and N values during division.
 |
| FIGURE 1 Mitosis |
It is possible to visualize the process of DNA synthesis within either nuclei or chromosomes by the incorporation of a radioactive precursor to DNA into cells and subsequent detection by autoradiography. Incorporation of thymidine, a DNA precursor, will only occur during the S phase, and not during G1 nor G2. If a pulse (short period of exposure) of 3H-thymidine is presented to cells, those that are in the S-period will incorporate this radioactive substance, while all others will not. Careful application of the pulse will allow the calculation of the duration of the S-phase. By knowing the timing for the entire cycle (from mitosis to mitosis), one can deduce the G1 and G2 periods.
Meiotic division differs from mitosis in that there are 2 division cycles instead of 1. In the first cycle, interphase is the same as for mitosis. That is, there is an S-phase with corresponding G1 and G2. During the second interphase, however, no DNA synthesis occurs. Consequently, there is no G1 or G2 in the second interphase. The result is that chromosomes are replicated prior to meiosis, and do not replicate again during meiosis.
Prophase
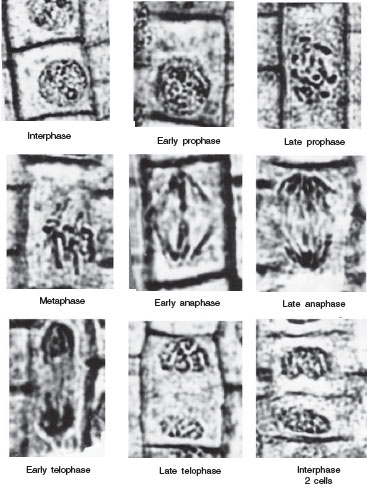
The first phase of mitosis is marked by the early condensation of the chromosomes into visible structures. At first, the chromatids are barely visible, but as they continue to coil, the chromosomes become thicker and shorter. The nuclear envelope is still present during this stage, as are any nucleolar structures. The centrioles are moving to the poles of the cell and spindle fibers are just beginning to form.
 |
| FIGURE 2 |
Metaphase
During the middle phase of karyokinesis, the chromosomes line up in the center of the cell, and form a metaphase plate. Viewed on edge, the chromosomes appear to be aligned across the entire cell, but viewed from 90° they appear to be spread throughout the entire cell (visualize a plate from its edge or from above). Each chromosome has a clear primary constriction, the centromere, and attached to each is a definitive spindle fiber. The spindle apparatus is completely formed, and the centrioles have reached their respective poles. The nucleolus and the nuclear envelope have disappeared.
Anaphase
The movement phase begins precisely as the 2 halves of a chromosome, the chromatids, separate and begin moving to the opposite poles. The centromere will lead the way in this process, and the chromatids form a V with the centromeres pointing toward the respective poles.
Telophase
The last phase is identified by the aggregation of the chromatids (now known as chromosomes) at the respective poles. During this phase, the chromosomes uncoil, the nuclear envelope is resynthesized, the spindle apparatus is dismantled, and the nucleolus begins to appear.
Meiosis
For meiosis, the phases prophase, metaphase, anaphase, and telophase are identified, but because there are 2 divisions, there are 2 sets. These are designated by Roman numerals; thus, prophase I, metaphase I, anaphase I, telophase I, interphase, prophase II, metaphase II, anaphase II, and telophase II. Interphase is normally not designated with a Roman numeral. Because of the significance of the chromosome pairing that occurs in prophase I, it is further subdivided into stages.
The phases of prophase I are named for the appearance of a thread-like structure, known as nema. Leptonema means “thin thread” and leptotene is the adjective applied to the term stage, i.e., proper terminology is the “leptotene stage” of prophase I. The word stage is often omitted.
Prophase I: Leptotene
This stage is marked by the first appearance of the chromosomes when they are in their most extended form (except for during interphase). They appear as a string with beads. The beads are known as chromomeres. The chromatids have already replicated prior to this phase, but typically, the replicated chromatids cannot be observed during the leptotene stage.
Prophase I: Zygotene
Zygos means “yoked,” and during this stage, the homologous chromosomes are seen as paired units. The chromosomes are shorter and thicker than in leptotene, and in some cells they remain attached to the nuclear envelope at the points near the aster. This gives rise to an image termed the “bouquet.” This attachment is rare in invertebrates and absent in plants, where the chromosomes appear to be a tangled mass.
Prophase I: Pachytene
When the pairing of zygotenes is complete, the chromosomes appear as thick strings, or pachynema. The chromosomes are about ¼ the length they were in leptotene, and there are obviously 2 chromosomes, with 2 chromatids in each bundle. The 2 chromosomes are referred to as a “bivalent,” while the same structure viewed as 4 chromatids is known as a “tetrad.”
Prophase I: Diplotene
This stage results as the gap between the 2 homologous chromosomes widens. The homologs have already paired during zygotene, recombined during pachytene, and are now beginning to repel each other. During this stage, the chromosomes of some species uncoil somewhat, reversing the normal direction typical of prophase. As the chromosomes separate, they are observed to remain attached at points known as “chiasmata.” These are believed to be the locations where genetic recombination of the genes has taken place.
Prophase I: Diakinesis
Prophase I ends as the homologs completely repel each other. The chromosomes will continue to coil tightly (reversing the slight uncoiling of the diplotene) and will reach their greatest state of contraction. As diakinesis progresses, chiasmata appear to move toward the ends of the chromosomes, a process known as “terminalization.” Since this stage is the end of prophase, the nucleolus usually disappears, along with the nuclear envelope.
Metaphase I
The tetrads move toward the center and line up on a metaphase plate. The nuclear envelope completely disappears. As the tetrads align themselves in the middle of the cell, they attach to spindle fibers in a unique manner. The centromeres of a given homolog will attach to the spindles from only 1 pole.
Anaphase I
The unique event occurring at this phase is the separation of the homologs. In contrast to mitotic anaphase, the centromeres of a given homolog do not divide, and consequently each homolog moves toward opposite poles. This results in a halving of the number of chromosomes, and is the basis of the reduction division that characterizes meiosis.
Telophase I: Interphase
Telophase in meiosis is similar to that of mitosis, except that in many species, the chromosomes do not completely uncoil. If the chromosomes do uncoil and enter a brief interphase, there is no replication of the chromatids. Remember that the chromatids have already been replicated prior to prophase I.
Prophase II: Telophase II
These phases are essentially identical in meiotic and mitotic division: the only distinction is that the chromosome number is half of that prior to meiosis. Each chromosome (homolog) is composed of 2 chromatids, and during anaphase II, the 2 chromatids of each chromosome move apart and become separate chromosomes.
While the 2 chromatids remain attached at the centromere, they are known as chromatids. Immediately upon separating, each chromatid becomes known as a chromosome and is no longer referred to as a chromatid. This is the reason that a cell can divide 1 chromosome (with 2 chromatids) into 2 cells, each with a chromosome—the term applied to the chromatid is changed.
Damage Induced During Division
In 1949, Albert Levan developed what was to become known as the Allium test for chromosome damage. Growing roots from onion bulbs were soaked in various agents and analyzed for their effect on mitosis. It was discovered that caffeine, for example, caused complete inhibition of mitosis, primarily through the inhibition of cell plate formation.
This test was later used extensively by B.A. Kihlman and extended to other higher plants. Kihlman found that 1 to 24 hour treatments of cells with caffeine and related oxypurines not only inhibited mitosis, but induced significant chromosome alterations (aberrations). Specifically, this treatment induced “stickiness” and “pseudochiasmata.” Stickiness is the clumping of chromosomes at metaphase and the formation of chromatin bridges at anaphase. Pseudochiasmata is the formation of side-arm bridges during anaphase. Caffeine also causes the formation of other chromosome and chromatid breaks and exchanges.
Colchicine, a drug that inhibits spindle fiber formation during mitosis, can be added to the growing cells to halt cell division at metaphase. This often will result in a doubling of the chromosome number, since colchicine typically inhibits cytokinesis, but not karyokinesis. The doubled chromosomes will fuse within a single nucleus, thus increasing the ploidy value of the nucleus. Moreover, methylated oxypurines (caffeine, theophylline, 8-ethoxycaffeine) are inhibitors of cell plate formation. Treatment with these agents for 0.5–1 hour, with concentrations as low as 0.02%–0.04%, results in the cell’s failure to undergo cytokinesis; in addition, the nuclei do not fuse into a single unit.
Thus, treatment with any of these agents should result in binucleate or multinucleate cells. In addition, alkylating agents such as (di-chloroethyl)methylamine or nitrogen mustard, di-epoxypropyl ether (DEPE) and b-Propiolactone (BPL), nitrosocompounds (N-Nitroso-N-methylurethan (NMU)), N-Methyl-phenylnitrosamine (MPNA), N-hydroxylphenylnitrosamine-ammonium (cupferron), and 1-Methyl-3-nitrosoguanidine (MNNG) have all been indicated as potent chromosome-breaking agents. Other compounds have included such things as maleic hydrazide, potassium cyanide, hydroxylamine, and dyes such as acridine orange in visible light. The damages involve abnormal metaphases, isochromatid breaks, chromatid exchanges, and anaphase bridges, to name a few.




