Biochemical Test for Identification of Bacteria
IntroductionStaining provides valuable information about bacterial morphology, Gram reaction, and presence of such structures as capsules and endospores. Beyond that, however, microscopic observation provides little additional information as to the genus and species of a particular bacterium.
To identify bacteria, we must rely heavily on biochemical testing. The types of biochemical reactions each organism undergoes act as a “thumbprint” for its identification. This is based on the following chain of logic:
- Each different species of bacterium has a different molecule of DNA (i.e., DNA with a unique series of nucleotide bases).
- Since DNA codes for protein synthesis, then different species of bacteria must, by way of their unique DNA, be able to synthesize different protein enzymes.
- Enzymes catalyze all of the various chemical reactions of which the organism is capable. This, in turn, means that different species of bacteria must carry out different and unique sets of biochemical reactions.
When identifying a suspected organism, you inoculate a series of differential media. After incubation, you then observe each medium to see if specific end products of metabolism are present. This can be done by adding indicators to the medium that react specifically with the end product being tested, producing some form of visible reaction, such as a color change. The results of these tests on the suspected microorganism are then compared to known results for that organism to confirm its identification.
Different bacteria, because of their unique enzymes, are capable of different biochemical reactions. Biochemical testing will also show the results of the activity of those enzymes. In later labs, we will use a wide variety of specialpurpose differential media frequently used in the clinical laboratory to identify specific pathogenic and opportunistic bacteria.
In general, we can classify enzymes as being either exoenzymes or endoenzymes. Exoenzymes are secreted by bacteria into the surrounding environment in order to break down larger nutrient molecules so they may enter the bacterium. Once inside the organism, some of the nutrients are further broken down to yield energy for driving various cellular functions, while others are used to form building blocks for the synthesis of cellular components. These later reactions are catalyzed by endoenzymes located within the bacterium.
Starch Hydrolysis
Introduction
Starch is a polysaccharide, which appears as a branched polymer of the simple sugar glucose. This means that starch is really a series of glucose molecules hooked together to form a long chain. Additional glucose molecules then branch off of this chain as shown below: GLU
|
( —GLU-GLU-GLU-GLU-GLU-GLU-GLU— )n Some bacteria are capable of using starch as a source of carbohydrate, but in order to do this, they must first hydrolyze or break down the starch so it may enter the cell. The bacterium secretes an exoenzyme, which hydrolyzes the starch by breaking the bonds between the glucose molecules. This enzyme is called a diastase.
—GLU / GLU / GLU / GLU / GLU / GLU / GLU— )n
action of diastase The glucose can then enter the bacterium and be used for metabolism.
Medium and organisms: Trypticase soy broth cultures of Bacillus subtilis and Escherichia coli.
Procedure
- Using a wax marker, draw a line on the bottom of a starch agar plate so as to divide the plate in half. Label one half B. subtilis and the other half E. coli.
- Make a single streak line, with the appropriate organism on the corresponding half of the plate.
- Incubate at 37°C until the next lab period.
- Next period, iodine will be added to see if the starch remains in the agar or has been hydrolyzed by the exoenzyme diastase. Iodine reacts with starch to produce a dark brown or blue/black color. If starch has been hydrolyzed, there will be a clear zone around the bacterial growth. If starch has not been hydrolyzed, the agar will remain a dark brown or blue/black color.
Protein Hydrolysis
Introduction
Proteins are made up of various amino acids linked together in long chains by means of peptide bonds. Many bacteria can hydrolyze a variety of proteins into peptides (short chains of amino acids) and eventually into individual amino acids. They can then use these amino acids to synthesize their own proteins and other cellular molecules, or to obtain energy. The hydrolysis of protein is termed proteolysis and the enzyme involved is called a protease. In this exercise we will test for bacterial hydrolysis of the protein casein, the protein that gives milk its white, opaque appearance.
Organisms: Trypticase soy broth cultures of Bacillus subtilis and Escherichia coli.
Procedure
- Divide the skim milk agar plate in half and inoculate one half with Bacillus subtilis and the other half with Escherichia coli, as done above with the above starch agar plate.
- Incubate at 37°C until the next lab period. If casein is hydrolyzed, there will be a clear zone around the bacterial growth. If casein is not hydrolyzed, the agar will remain white and opaque.
Fermentation of Carbohydrates
Introduction
Carbohydrates are complex chemical substrates, which serve as energy sources when broken down by bacteria and other cells. They are composed of carbon, hydrogen, and oxygen (with hydrogen and oxygen in the same ratio as water; [CH2O]) and are usually classed as either sugars or starches.
Facultative anaerobic and anaerobic bacteria are capable of fermentation, an anaerobic process during which carbohydrates are broken down for energy production. A wide variety of carbohydrates may be fermented by various bacteria in order to obtain energy. The types of carbohydrates which are fermented by a specific organism can serve as a diagnostic tool for the identification of that organism.
We can detect whether a specific carbohydrate is fermented by looking for common end products of fermentation.
When carbohydrates are fermented as a result of bacterial enzymes, the following fermentation end products may be produced:
- Acid end products, or
- Acid and gas end products.
In order to test for these fermentation products, inoculate and incubate tubes of media containing a single carbohydrate (such as lactose or maltose), a pH indicator (such as phenol red), and a durham tube (a small inverted tube to detect gas production). If the particular carbohydrate is fermented by the bacterium, acid end products will be produced, which lower the pH, causing the pH indicator to change color (phenol red turns yellow). If gas is produced along with the acid, it collects in the durham tube as a gas bubble. If the carbohydrate is not fermented, no acid or gas will be produced and the phenol red will remain red.
Media: 3 tubes of phenol red lactose broth and 3 tubes of phenol red maltose broth.
Organisms: Trypticase soy agar cultures of Bacillus subtilis, Escherichia coli, and Staphylococcus aureus.
Procedure
- Label each tube with the name of the sugar in the tube and the name of the bacterium you are growing.
- Inoculate 1 phenol red lactose broth tube and 1 phenol red maltose broth tube with Bacillus subtilis.
- Inoculate a second phenol red lactose broth tube and a second phenol red maltose broth tube with Escherichia coli.
- Inoculate a third phenol red lactose broth tube and a third phenol red maltose broth tube with Staphylococcus aureus.
- Incubate all tubes at 37°C until next lab period
Indole and Hydrogen Sulfide Production
Introduction
Sometimes we look for the production of products produced by only a few bacteria. As an example, some bacteria use the enzyme tryptophanase to convert the amino acid tryptophan into molecules of indole, pyruvic acid, and ammonia. Since only a few bacteria contain tryptophanase, the formation of indole from a tryptophan substrate can be another useful diagnostic tool for the identification of an organism. Indole production is a key test for the identification of Escherichia coli.
By adding Kovac’s reagent to the medium after incubation, we can determine if indole was produced. Kovac’s reagent will react with the indole and turn red.
Likewise, some bacteria are capable of breaking down sulfur-containing amino acids (cystine, methionine) or reducing inorganic sulfur-containing compounds (such as sulfite, sulfate, or thiosulfate) to produce hydrogen sulfide (H2S). This reduced sulfur may then be incorporated into other cellular amino acids, or perhaps into coenzymes. The ability of an organism to reduce sulfurcontaining compounds to hydrogen sulfide can be another test for identifying unknown organisms, such as certain Proteus and Salmonella. To test for hydrogen sulfide production, a medium with a sulfur-containing compound and iron salts is inoculated and incubated. If the sulfur is reduced and hydrogen sulfide is produced, it will combine with the iron salt to form a visible black ferric sulfide (FeS) in the tube.
Medium: Three tubes of SIM (Sulfide, Indole, Motility) medium. This medium contains a sulfur source, an iron salt, the amino acid tryptophan, and is semisolid in agar content (0.3%). It can be used to detect hydrogen sulfide production, indole production, and motility.
Organisms: Trypticase soy agar cultures of Proteus mirabilis, Escherichia coli, and Enterobacter cloacae.
Procedure
- Stab a SIM medium tube with Proteus mirabilis.
- Stab a second SIM medium tube with Escherichia coli.
- Stab a third SIM medium tube with Enterobacter cloacae.
- Incubate at 37°C until the next lab period.
- Next lab period, add Kovac’s reagent to each tube to detect indole production.
Catalase Activity
Introduction
Catalase is the name of an enzyme found in most bacteria, which initiates the breakdown of hydrogen peroxide (H2O2) into water (H2O) and free oxygen (O2).
During the normal process of aerobic respiration, hydrogen ions (H+) are given off and must be removed by the cell. The electron transport chain takes these hydrogen ions and combines them with half a molecule of oxygen (an oxygen atom) to form water (H2O). During the process, energy is given off and is trapped and stored in ATP. Water is then a harmless end product. Some cytochromes in the electron transport system, however, form toxic hydrogen peroxide (H2O2) instead of water, and this must be removed. This is done by the enzyme catalase breaking the hydrogen peroxide into water and oxygen, as shown above. Most bacteria are catalase-positive; however, certain genera that don’t carry out aerobic respiration, such as Streptococcus, Lactobacillus, and Clostridium, are catalase-negative.
Materials
- Trypticase soy agar cultures of Staphylococcus aureus and Streptococcus lactis
- 3% hydrogen peroxide
Procedure
Add a few drops of 3% hydrogen peroxide to each culture and look for the release of oxygen as a result of hydrogen peroxide breakdown. This appears as foaming.
Results of a Biochemical Tests
Starch Hydrolysis
When iodine is added to starch, the iodine-starch complex that forms produces a characteristic dark brown or deep purple color reaction. If the starch has been hydrolyzed into glucose molecules by the diastase exoenzyme, it no onger produces this reaction. Therefore, flood the surface of the starch agar plate with Gram’s iodine. If the exoenzymes of the organism broke down the starch in the agar, a clear zone will surround the bacterial growth. If the organism lacks the exoenzyme to break down the starch, the agar around the growth should turn dark brown or blue/black due to the iodine-starch complex.
Record your results and indicate which organism was capable of hydrolyzing the starch (+ = hydrolysis; – = no hydrolysis).
Protein Hydrolysis
The protein casein exists as a colloidal suspension in milk and gives milk its characteristic white, opaque appearance. If the casein in the milk is hydrolyzed into peptides and amino acids, it will lose its opaqueness. Therefore, if the bacteria has the exoenzyme capable of casein hydrolysis, there will be a clear zone around the bacterial growth. If the organism lacks the exoenzyme to break down casein, the skim milk agar will remain white and opaque.
Record your results and indicate which organism was capable of hydrolyzing casein (+ = hydrolysis; – = no hydrolysis).
Fermentation of Carbohydrates
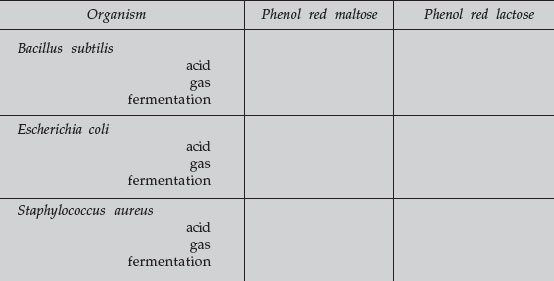
Phenol red pH indicator is red at neutral pH and yellow at acid pH. A change in color in the tube from red to yellow indicates that the organism has fermented that particular carbohydrate, producing acid end products. Gas bubbles in the durham tube indicate gas was also produced from fermentation. (The results of fermentation may be acid alone or acid plus gas, but never gas alone.) If the phenol red remains red, no acid was produced and the carbohydrate was not fermented.
- Carbohydrate fermentation producing acid but no gas.
- Carbohydrate fermentation producing acid and gas.
- No carbohydrate fermentation. No acid or gas.
Carbohydrate Fermentation
Record your results below (+ = positive; – = negative).
Production of Indole and Hydrogen Sulfide
- Observe the SIM tubes. A black color indicates the organism has produced hydrogen sulfide.
- Carefully add a dropper full of Kovac’s reagent to each tube. A red color indicates the production of indole.
SIM Medium
Record your results below (+ = positive; – = negative).
 |
 |
Figure 39 |
Catalase Activity
If the bacterium produces the enzyme catalase, then the hydrogen peroxide added to the culture will be broken down into water and free oxygen. The oxygen will bubble through the water causing a surface froth to form. A catalasenegative bacterium will not be able to break down the hydrogen peroxide, and no frothing will occur.
Catalase Test
Record your results (foaming = positive; no foaming = negative).
Performance Objectives
Introduction
- Explain the chemical nature and function of enzymes.
- Define endoenzyme and exoenzyme.
Starch Hydrolysis
Discussion
Describe a method of testing for starch hydrolysis and how to interpret the results.
Results
Interpret the results of starch hydrolysis on a starch agar plate that has been inoculated, incubated, and flooded with iodine.
Protein Hydrolysis
Discussion
Describe a method for testing casein hydrolysis and how to interpret the results.
Results
Interpret the results of casein hydrolysis on a skim milk agar plate after it has been inoculated and incubated.
Fermentation of Carbohydrates
Discussion
Name the general end products that may be formed as a result of the bacterial fermentation of sugars, and describe how these end products change the appearance of a broth tube containing a sugar, the pH indicator phenol red, and a durham tube.
Results
Interpret the carbohydrate fermentation results in tubes of phenol red carbohydrate broth containing a durham tube after it has been inoculated and incubated.
Indole and Hydrogen Sulfide Production
Discussion
- Name the pathway for the breakdown of tryptophan to indole.
- Name the pathway for the detection of sulfur reduction in SIM medium.
- Describe 3 reactions that may be tested for in SIM medium, and how to interpret the results.
Results
Interpret the hydrogen sulfide and indole results in a SIM medium tube after inoculation, incubation, and addition of Kovac’s reagent.
Catalase Activity
Discussion
Describe the function of the enzyme catalase, and a method of testing for catalase activity.
Results
Interpret the results of a catalase test after adding hydrogen peroxide to a plate culture of bacteria.




