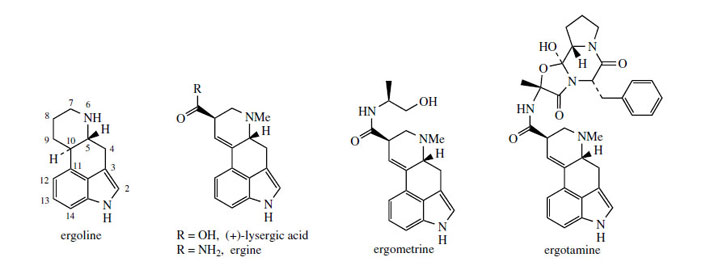
Ergot Alkaloids
Ergot* is a fungal disease commonly found on
many wild and cultivated grasses, and is caused
by species of Claviceps. The disease is eventually
characterized by the formation of hard, seedlike
‘ergots’ instead of normal seeds, these structures, called sclerotia, forming the resting stage of the
fungus. The poisonous properties of ergots in grain,
especially rye, for human or animal consumption
have long been recognized, and the causative
agents are known to be a group of indole alkaloids,
referred to collectively as the ergot alkaloids
or ergolines (Figure 99). Under natural conditions
the alkaloids are elaborated by a combination
of fungal and plant metabolism, but they can be synthesized in cultures of suitable Clavicepsspecies. Ergoline alkaloids have also been found
in fungi belonging to genera Aspergillus, Rhizopus, and Penicillium, as well as Claviceps, and
simple examples are also found in some plants of
the Convolvulaceae such as Ipomoea and Rivea(morning glories)*. |
Despite their toxicity, some of these alkaloids have valuable pharmacological activities and are used clinically on a routine basis.
 |
| Figure 99 |
 |
| Figure 100 |
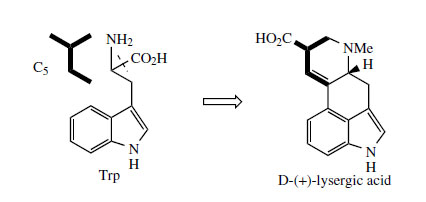
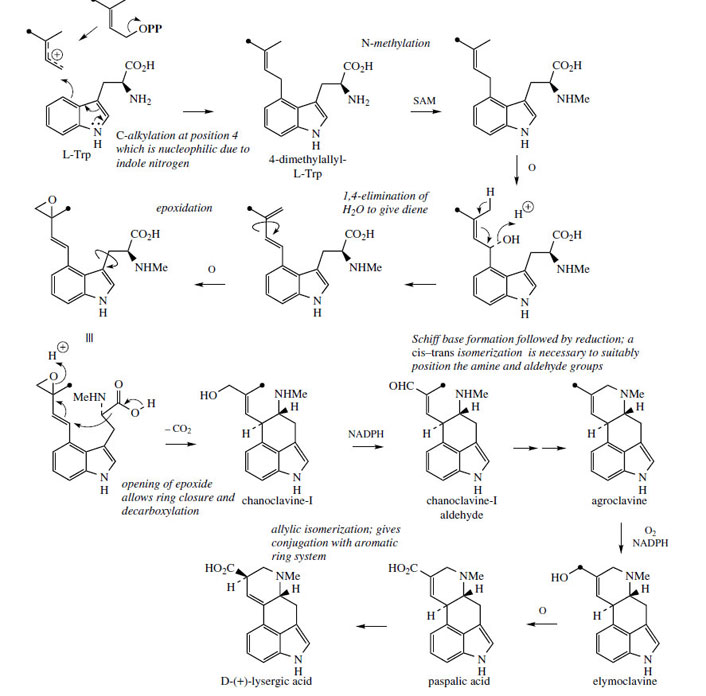
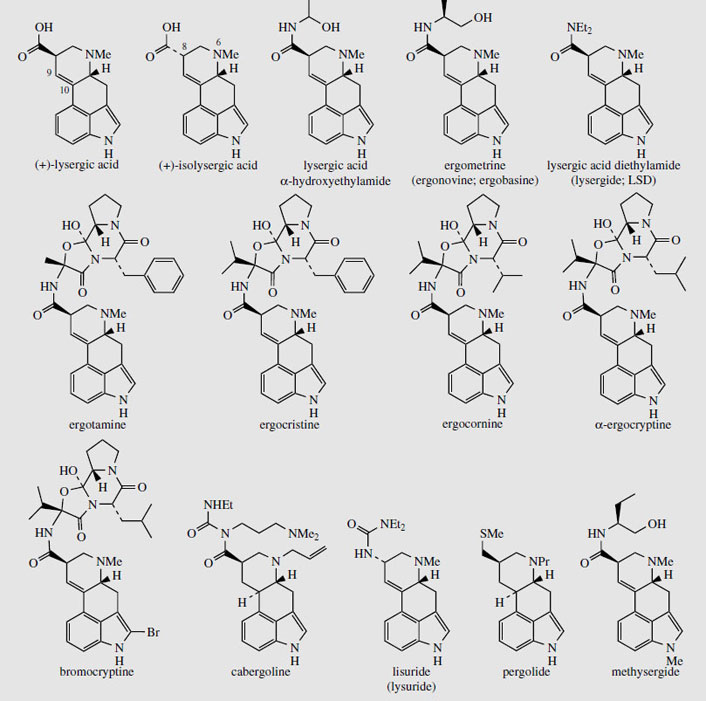
Medicinally useful alkaloids are derivatives of (+)-lysergic acid (Figure 99), which is typically bound as an amide with an amino alcohol as in ergometrine, or with a small polypeptide structure as in ergotamine. The building blocks for lysergic acid are tryptophan (less the carboxyl group) and an isoprene unit (Figure 100). Alkylation of tryptophan with dimethylallyl diphosphate gives 4-dimethylallyl-Ltryptophan, which then undergoes N-methylation (Figure 101). Formation of the tetracyclic ring system of lysergic acid is known to proceed through chanoclavine-I and agroclavine, though the mechanistic details are far from clear. Labelling studies have established that the double bond in the dimethylallyl substituent must become a single bond on two separate occasions, allowing rotation to occur as new rings are established. This gives the appearance of cis–trans isomerizations as 4-dimethylallyl- L-tryptophan is transformed into chanoclavine- I, and as chanoclavine-I aldehyde cyclizes to agroclavine (Figure 101). A suggested sequence to account for the first of these is shown. In the later stages, agroclavine is hydroxylated to elymoclavine, further oxidation of the primary alcohol occurs giving paspalic acid, and lysergicacid then results from a spontaneous allylic isomerization.
 |
| Figure 101 |
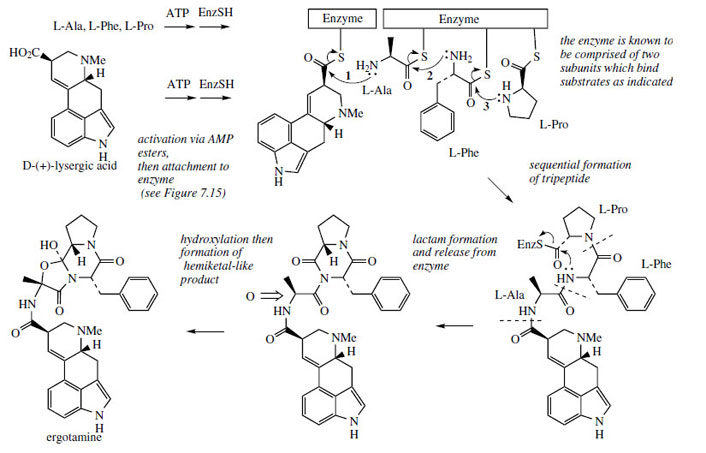
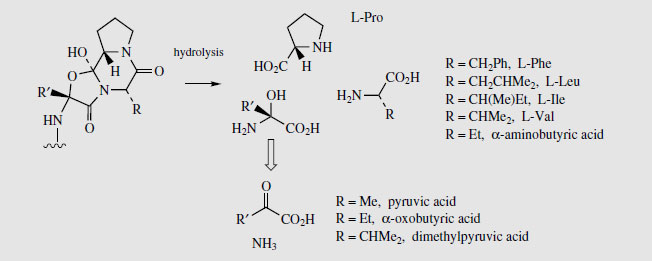
Simple derivatives of lysergic acid require the formation of amides; for example, ergine (Figure 99) in Rivea and Ipomoea species is lysergic acid amide, whilst ergometrine from Claviceps purpurea is the amide with 2-aminopropanol. The more complex structures containing peptide fragments, e.g. ergotamine (Figure 99), are formed by sequentially adding amino acid residues to thioester-bound lysergic acid, giving a linear lysergyl–tripeptide covalently attached to the enzyme complex (Figure 102). Peptide formation involves the same processes seen in the nonribosomal biosynthesis of peptides, and involves ATP-mediated activation of the amino acids prior to attachment to the enzyme complex through thioester linkages. A phosphopantatheine arm is used to enable the growing chain to reach the various active sites (compare fatty acid biosynthesis, page 36). The cyclized tripeptide residue is readily rationalized by the formation of a lactam (amide) which releases the product from the enzyme, followed by generation of a hemiketal-like linkage as shown (Figure 102).
 |
| Figure 102 |
Ergot
Medicinal ergot is the dried sclerotium of the fungus Claviceps purpurea (Clavicipitaceae) developed on the ovary of rye, Secale cereale (Graminae/Poaceae). Ergot is a fungal disease of wild and cultivated grasses, and initially affects the flowers. In due course, a dark sclerotium, the resting stage of the fungus, is developed instead of the normal seed. This protrudes from the seed head, the name ergot being derived from the French word argot - a spur. The sclerotia fall to the ground, germinating in the spring and reinfecting grasses or grain crops by means of spores. Two types of spore are recognized: ascospores, which are formed in the early stages and are dispersed by the wind, whilst later on conidiospores are produced, which are insect distributed. The flowers are only susceptible to infection before pollination. Ergots may subsequently be harvested with the grain and contaminate flour or animal feed. The consumption of ergot-infected rye has resulted in the disease ergotism, which has a long, well documented history.
There are three broad clinical features of ergot poisoning, which are due to the alkaloids present and the relative proportions of each component:
| - | Alimentary upsets, e.g. diarrhoea, abdominal pains, and vomiting. |
| - | Circulatory changes, e.g. coldness of hands and feet due to a vasoconstrictor effect, a decrease in the diameter of blood vessels, especially those supplying the extremeties. |
| - | Neurological symptoms, e.g. headache, vertigo, convulsions, psychotic disturbances, and hallucinations. |
These effects usually disappear on removal of the source of poisoning, but much more serious problems develop with continued ingestion, or with heavy doses of ergot-contaminated food. The vasoconstrictor effect leads to restricted blood flow in small terminal arteries, death of the tissue, the development of gangrene, and even the shedding of hands, feet, or limbs. Gangrenous ergotism was known as St Anthony's Fire, the Order of St Anthony traditionally caring for sufferers in the Middle Ages. The neurological effects were usually manifested by severe and painful convulsions. Outbreaks of the disease in both humans and animals were relatively frequent in Europe in the Middle Ages, but once the cause had been established it became relatively simple to avoid contamination. Separation of the ergots from grain, or the use of fungicides during cultivation of the crop, have removed most of the risks, though infection of crops is still common.
 |
| Figure 103 |
 |
| Table 6.1 |
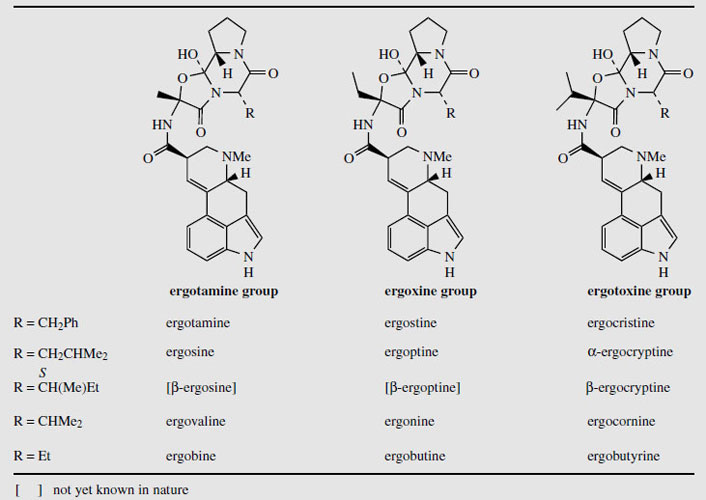
The ergot sclerotia contain from 0.15-0.5% alkaloids, and more than 50 have been characterized. The medicinally useful compounds are derivatives of (+)-lysergic acid (Figure 103), and can be separated into two groups, the water-soluble amino alcohol derivatives (up to about 20% of the total alkaloids), and water-insoluble peptide derivatives (up to 80% of the total alkaloids). Ergometrine (Figure 103) (also known as ergonovine in the USA and ergobasine in Switzerland) is an amide of lysergic acid and 2-aminopropanol, and is the only significant member of the first group. The peptide derivatives contain a cyclized tripeptide fragment bonded to lysergic acid via an amide linkage. Based on the nature of the three amino acids, these structures can be subdivided into three groups, the ergotamine group, the ergoxine group, and the ergotoxine group (Table 6.1). The amino acids involved are alanine, valine, leucine, isoleucine, phenylalanine, proline, and α-aminobutyric acid, in various combinations (Figure 104). All contain proline in the tripeptide, and one of the amino acids is effectively incorporated into the final structure in the form of an a-hydroxy-a-amino acid. Thus, ergotamine incorporates alanine, phenylalanine, and proline residues in its peptide portion. Hydrolysis gives (+)&minuslysergic acid, proline, and phenylalanine, together with pyruvic acid and ammonia, the latter hydrolysis products a consequence of the additional hydroxylation involving alanine (Figure 104). Hydrolysis of the ergotoxine group of alkaloids results in the proximal valine unit being liberated as dimethylpyruvic acid (not systematic nomenclature) and ammonia, and the ergoxine group similarly yields α-oxobutyric acid from the α-aminobutyric acid fragment. The alkaloid 'ergotoxine' was originally thought to be a single compound, but was subsequently shown to be a mixture of alkaloids. The proposed structures β-ergosine and β-ergoptine, which complete the combinations shown in Table 6.1, have not yet been isolated as natural products.
 |
| Figure 104 |
Medicinal ergot is cultivated in the Czech Republic, Germany, Hungary, Switzerland, Austria, and Poland. Fields of rye are infected artificially with spore cultures of Claviceps purpurea, either by spraying or by a mechanical process that uses needles dipped in a spore suspension. The ergots are harvested by hand, by machine, or by separation from the ripe grain by flotation in a brine solution. By varying the strain of the fungal cultures, it is possible to maximize alkaloid production (0.4-1.2%), or give alkaloid mixtures in which particular components predominate. Ergots containing principally ergotamine in concentrations of about 0.35% can be cultivated. In recent times, ergot of wheat (Triticum aestivum), and the wheat-rye hybrid triticale (Triticosecale) have been produced commercially.
Alternatively, the ergot alkaloids can be produced by culturing the fungus. Initially, cultures of the rye parasite Claviceps purpurea in fermentors did not give the typical alkaloids associated with the sclerotia, e.g. ergometrine and ergotamine. These medicinally useful compounds appear to be produced only in the later stages of development of the fungus. Instead, the cultures produced alkaloids that were not based on lysergic acid, and are now recognized as intermediates in the biosynthesis of lysergic acid, e.g. chanoclavine- I, agroclavine, and elymoclavine (Figure 101). Ergot alkaloids that do not yield lysergic acid on hydrolysis have been termed clavine alkaloids. Useful derivatives based on lysergic acid can be obtained by fermentation growth of another fungal species, namely Claviceps paspali. Although some strains are available that produce peptide alkaloids in culture, other strains produce high yields of simple lysergic acid derivatives. These include lysergic acid α-hydroxyethylamide (Figure 103), lysergic acid amide (ergine) (Figure 99), which is also an acid-catalysed decomposition product from lysergic acid α-hydroxyethylamide, and the Δ8,9-isomer of lysergic acid, paspalic acid (Figure 101). Lysergic acid is obtained from the first two by hydrolysis, or from paspalic acid by allylic isomerization. Other alkaloids, e.g. ergometrine and ergotamine, can then be produced semi-synthetically. High yielding fermentation methods have also been developed for direct production of ergotamine and the ergotoxine group of peptide alkaloids.
The pharmacologically active ergot alkaloids are based on (+)-lysergic acid (Figure 103), but since one of the chiral centres in this compound (and its amide derivatives) is adjacent to a carbonyl, the configuration at this centre can be changed as a result of enolization brought about by heat or base (compare tropane alkaloids, page 298; again note that enolization is favoured by conjugation with the aromatic ring). The new diastereomeric form of (+)- lysergic acid is (+)-isolysergic acid (Figure 103), and alkaloids based on this compound are effectively pharmacologically inactive. They are frequently found along with the (+)-lysergic acid derivatives, amounts being significant if old ergot samples are processed, or unsuitable isolation techniques are employed. In the biologically active lysergic acid derivatives, the amide group occupies an 8-equatorial position, whilst in the inactive iso-forms, this group is axial. However, the axial form is actually favoured because this configuration allows hydrogen-bonding from the amide N-H to the hetrocyclic nitrogen at position 6. Derivatives of (+)-isolysergic acid are named by adding the syllable -in- to the corresponding (+)-lysergic acid compound, e.g. ergometrinine, ergotaminine.
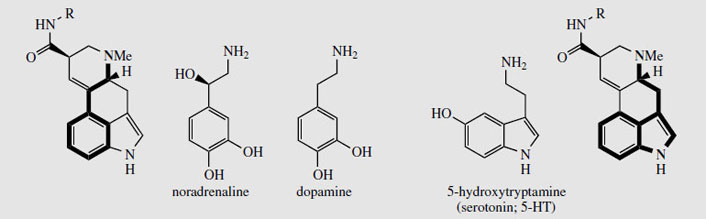
The ergot alkaloids owe their pharmacological activity to their ability to act at a-adrenergic, dopaminergic and serotonergic receptors. The relationship of the general alkaloid structure to those of noradrenaline, dopamine, and 5-hydroxytryptamine (5-HT, serotonin) is shown in Figure 105. The pharmacological response may be complex. It depends on the preferred receptor to which the compound binds, though all may be at least partially involved, and whether the alkaloid is an agonist or antagonist.
 |
| Figure 105 |
Despite the unpleasant effects of ergot as manifested by St Anthony's Fire, whole ergot preparations have been used since the 16th century to induce uterine contractions during childbirth, and to reduce haemorrhage following the birth. This oxytocic effect (oxytocin is the pituitary hormone that stimulates uterine muscle) is still medicinally valuable, but is now achieved through use of the isolated alkaloid ergometrine. The deliberate use of ergot to achieve abortions is dangerous and has led to fatalities.
Ergometrine (ergonovine) (Figure 103) is used as an oxytocic, and is injected during the final stages of labour and immediately following childbirth, especially if haemorrhage occurs. Bleeding is reduced because of its vasoconstrictor effects, and it is valuable after Caesarian operations. It is sometimes administered in combination with oxytocin itself. Ergometrine is also orally active. It produces faster stimulation of uterine muscle than do the other ergot alkaloids, and probably exerts its effect by acting on a-adrenergic receptors, though it may also stimulate 5-HT receptors.
Ergotamine (Figure 103) is a partial agonist of α-adrenoceptors and 5-HT receptors. It is not suitable for obstetric use because it also produces a pronounced peripheral vasoconstrictor action. This property is exploited in the treatment of acute attacks of migraine, where it reverses the dilatation of cranial blood vessels. Ergotamine is effective orally, or by inhalation in aerosol form, and may be combined with caffeine, which is believed to enhance its action. The semi-synthetic dihydroergotamine is produced by hydrogenation of the lysergic acid Δ9,10 double bond (giving C-10 stereochemistry as in ergoline) and is claimed to produce fewer side-effects, especially digestive upsets.
The 'ergotoxine' alkaloid mixture also has oxytocic and vasoconstrictor activity but is only employed medicinally as the 9,10-dihydro derivatives dihydroergotoxine (co-dergocrine), a mixture in equal proportions of dihydroergocornine, dihydroergocristine, and the dihydroergocryptines (α- and β- in the ratio 2:1). In the case of these alkaloids, reduction of the double bond appears to reverse the vasoconstrictor effect, and dihydroergotoxine has a cerebral vasodilator activity. The increased blood flow is of benefit in some cases of senility and mild dementia, and helps to improve both mental function and physical performance.
A number of semi-synthetic lysergic acid derivatives act by stimulation of dopamine receptors in the brain, and are of value in the treatment of neurological disorders such as Parkinson's disease. Bromocriptine (2-bromo-α-ergocryptine), cabergoline, lisuride (lysuride), and pergolide(Figure 103) are all used in this way. Bromocriptine and cabergoline find wider use in that they also inhibit release of prolactin by the pituitary, and can thus suppress lactation and be used in the treatment of breast tumours. Methysergide (Figure 103) is a semi-synthetic analogue of ergometrine, having a modified amino alcohol side-chain and an N-methyl group on the indole ring. It is a potent 5-HT antagonist and as such is employed in the prophylaxis of severe migraine headaches, though its administration has to be very closely supervised.
Prolonged treatment with any of the ergot alkaloids is undesirable and it is vital that the clinical features associated with ergot poisoning are recognized. Treatment must be withdrawn immediately if any numbness or tingling develops in the fingers or toes. Sideeffects will disappear on withdrawal of the drug, but there have been many cases where misdiagnosis has unfortunately led to foot or toe rot, and the necessity for amputation of the dead tissue.
Undoubtedly the most notorious of the lysergic acid derivatives is lysergide (lysergic acid diethylamide or LSD) (Figure 103). This widely abused hallucinogen, known as 'acid', is probably the most active and specific psychotomimetic known, and is a mixed agonist-anatagonist at 5-HT receptors, interfering with the normal processes. An effective oral dose is from 30 to 50 �g. It was synthesized from lysergic acid, and even the trace amounts absorbed during its handling were sufficient to give its creator quite dramatic hallucinations. LSD intensifies perceptions and distorts them. How the mind is affected depends on how the user is feeling at the time, and no two 'trips' are alike. Experiences can vary from beautiful visions to living nightmares, sometimes lasting for days. Although the drug is not addictive, it can lead to schizophrenia and there is danger of serious physical accidents occurring whilst the user is under the influence of the drug.
Morning Glories
Lysergic acid derivatives have also been characterized in the seeds of morning glory (Ipomoea violacea), Rivea corymbosa, and other members of the Convolvulaceae. Such seeds formed the ancient hallucinogenic drug ololiuqui still used by the Mexican Indians in religious and other ceremonies. Extracts from the ground seeds are swallowed and the narcotic and hallucinogenic effects are said to provide contact with the gods. The active constituent has been identified to be principally ergine (lysergic acid amide) (Figure 99), and this has an activity about one-20th that of LSD, but is more narcotic than hallucinogenic. The alkaloid content of the seeds is usually low, at about 0.05%, but higher levels (0.5-1.3%) have been recorded. Minor ergot-related constituents include ergometrine (Figure 103), lysergic acid α-hydroxyethylamide (Figure 103), the inactive isolysergic acid amide (erginine), and some clavine alkaloids.
Since morning glories are widely cultivated ornamentals and seeds are readily available, deliberate ingestion by thrill-seekers has been considerable. Although the biological activity is well below that of LSD, the practice is potentially dangerous.




