Purification of Clathrin-Coated Vesicles from Bovine Brain, Liver, and Adrenal Gland
I. INTRODUCTIONClathrin-coated vesicles are intermediates in several selective membrane transport processes in eukaryotic cells (for review, see Bonifacino and Lippincott- Schwartz, 2003; Brodsky et al., 2001; Schmid, 1997). They are derived from clathrin-coated membrane regions on the plasma membrane, the TGN, endosomes (Stoorvogel et al., 1996), and possibly also lysosomes (Traub et al., 1996) by a process of invagination and fission. In addition to the main structural protein clathrin, a three-legged molecule composed of one heavy and one light chain per leg, a number of other proteins have been isolated from clathrin-coated vesicles: most prominent are the adaptor protein (AP) complexes, heterotetramers consisting of two heavy chains, an intermediate chain, and a light chain (for review, see Kirchhausen, 1999). AP-1 and AP-2 complexes link clathrin to specific membrane areas by binding to clathrin N-terminal domains and to cytosolic tails of transmembrane receptors. The recently discovered AP-3 and AP-4 complexes appear to function independently of clathrin and are not enriched in clathrin-coated vesicles. Furthermore, clathrin-coated vesicles contain monomeric adaptor proteins, such as AP 180 and members of the epsin family. These interact with coat components via their poorly structured C-terminal regions (Kalthoff et al., 2002) and with phosphoinositides of the membrane via their N-terminal ENTH domain (for review, see De Camilli et al., 2002). Also present are accessory proteins such as auxilins that provide a J-like domain for the recruitment of Hsc70 to clathrin, thereby serving to initiate the removal of the clathrin coat (Ungewickell et al., 1995). A novel bilayered clathrin coat without curvature has been described on multivesicular endosomes (Raiborg et al., 2002; Sachse et al., 2002). It contains the monomeric clathrin adaptor protein HRS and appears to be involved in the sorting of ubiquitinated membrane proteins to internal vesicles. This flat structure probably does not to give rise to coated vesicles.
Since the first report on the purification of clathrincoated vesicles (Kanaseki and Kadota, 1969), a variety of protocols have been published. Most recent protocols are based on a purification procedure introduced by Campbell et al. (1984). This procedure rapidly provides crude clathrin-coated vesicles that are well suited for the preparation of various coat proteins (Ahle et al., 1988; Ahle and Ungewickell, 1990; Lindner and Ungewickell, 1991). This basic protocol has been further improved by the introduction of a centrifugation step involving a sucrose/D2O cushion (Maycox et al., 1992). By this step, contaminations of smooth membrane vesicles are reduced further. A similar procedure has been used to purify rat brain clathrincoated vesicles for proteomic characterization (Wasiak et al., 2002).
This article describes a purification protocol for bovine brain-coated vesicles that includes a sucrose/ D2O step gradient at the end. With appropriate volume adjustments, the protocol can also be used for the purification of coated vesicles from other species such as pigs or rats. For the preparation of clathrin-coated vesicles from other organs (adrenal gland, liver), some modifications in the protocol are suggested.
II. MATERIALS AND INSTRUMENTATION
Ficoll 400 (Cat. No. 17-0400-02) is from Pharmacia; sucrose (Cat. No. BP 220-1) is from Fisher Scientific; D2O (Cat. No. 15,188-2) is from Aldrich; EGTA (Cat. No. E-3889), MES (Cat. No. M-3023), and phenylmethylsulfonyl fluoride (PMSF, Cat. No. P-7626) are from Sigma. All other reagents are from Sigma or Fisher in analytical grade.
Biological material is obtained from an local abattoir within 1h of slaughter and is kept on ice until further processing (1-2h). Fresh and cleaned material can be frozen in liquid nitrogen and stored at -80°C for several months. It is helpful to cut the tissue into small pieces before freezing. This supports a rapid drop of temperature in the tissue and minimizes ice crystal formation. The latter process reduces yield and purity of the following preparation, probably due to the destruction of membrane vesicles and liberation of proteolytic activities.
For homogenization of the tissue, a Waring commercial blender (VWR International, Cat. No. 58977- 169) is used. Membrane pellets are resuspended with Potter-Elvehjem homogenizers of various sizes (10- 55ml) obtained from Fisher Scientific (Cat. No. 08414- 14 A to D). The metal shaft of the larger homogenizers is attached to a variable-speed overhead drive so that the pestle can be rotated.
Low-speed centrifugations are done in a Sorvall RC- 5B centrifuge using GS-3, GSA, or SS-34 heads. Highspeed centrifugations are performed with Beckman Ti 45, Ti 35, and SW 28 rotors in conventional ultracentrifuges. For fixed angle ultracentrifugation rotors, Beckman polycarbonate bottles with screw caps (Cat. No. 355622) are used and for the SW 28 rotor open-top ultraclear tubes (Cat. No. 344058) are used.
III. PROCEDURES
A. Cleaning of Bovine Brain Cortices
Solutions
Phosphate-buffered saline (PBS): 137mM NaCl, 2.7 mM KCl, 8.2mM Na2HPO4, 1.9 mM KH2PO4, 0.02% NaN3, pH 7.0. To make 1 liter of 10× PBS (stock solution), dissolve 80g NaCl, 2 g KCl, 2.58g KH2PO4, 11.64g Na2HPO4, and 0.2g NaN3 in double-distilled water. Adjust the pH to 7.0 (if necessary) and bring the volume to 1 liter. Dilute this stock solution 1:10 with double-distilled water and chill to 4°C prior to use. Approximately 2-3 liters of 1× PBS are needed per kilogram tissue.
Steps
- Separate the cerebellum and the lower part of the brain from the cortex.
- Take a hemisphere of the cortex, place it in an ice bucket covered with plastic wrap, and remove the meninges along with blood vessels contained therein using forceps.
- Collect the cleaned cortex hemispheres in a preweighed beaker on ice. Determine the weight of the tissue and estimate the volume of homogenization buffer needed in Section III,B (approximately 1 liter per kg of tissue).
- Wash the cortex hemispheres with cold PBS several times to remove the remaining blood. To do this, fill the beaker containing the hemispheres with PBS, mix gently, and pour the liquid off. It is helpful to use a household sieve at this step. Repeat the washes until the blood is removed.
- If you want to store the brain tissue for later processing, cut it into small pieces after the washing step and freeze it in liquid nitrogen. Store frozen material at -80°C. Otherwise continue with Section III,B.
B. Homogenization
Solutions
- Buffer A: 0.1M MES, 1.0mM EGTA, 0.5mM MgCl2, 0.02% NaN3, pH 6.5. To make 1 liter of 10x buffer A (stock solution), dissolve 195.2g MES, 3.8g EGTA, and 1.0g MgCl2 in double distilled water. Adjust the pH with 10N NaOH to 6.5, add 2 g NaN3, and bring the volume to 1 liter. Do not add NaN3 prior to the adjustment of the pH because MES is acidic and may release HN3. To prepare 1× buffer A for homogenization, dilute the stock solution 1:10 with doubledistilled water, chill to 4°C, and supplement with 0.1 mM PMSF prior to use. Prepare about 3 liters buffer A per kilogram tissue.
- Protease inhibitor (PMSF stock solution, 1000× = 0.1 M). To make 10 ml, dissolve 174 mg PMSF in 10 ml pure methanol, aliquot, and store at -20°C. Dilute 1: 1000 to get a working concentration of 0.1 mM. Note that PMSF will hydrolyze in water and that it is not soluble in this solvent at high concentrations.
Steps
- Fill the cup of a Waring commercial blender with 300-400 g of washed tissue and an equivalent amount of cold buffer A containing 0.1mM freshly added PMSE Do not fill the cup up to the top, but leave space below the rim.
- Homogenize the tissue by three to six bursts of 10-15s duration with the setting on maximum speed. The number of bursts required to give a good homogenization varies: for brain, usually three bursts are sufficient, whereas other organs, especially adrenal glands, require more (see Section IV). Do not increase the length of the bursts but their number to prevent heating of the homogenate.
C. Differential Centrifugations
1. Preparation of Postmitochondrial Supernatants
Steps
- Pour the homogenate into buckets of a Sorvall GS-3 or GSA rotor, balance the buckets, and centrifuge them in the precooled rotor at 7000rpm (about 8000g in both types of rotors) for 50min at 4°C.
- Pour the turbid supernatants through a funnel with several layers of gauze to separate floating lipids from the supernatant.
- The bulky pellets (up to a third of the total volume) should be resuspended with an at least equivalent volume of cold homogenization buffer and recentrifuged under the aforementioned conditions.
- Discard the pellets after the second centrifugation and keep the combined supernatants on ice.
2. Preparation of Microsomal Pellets
Steps
- Fill the postmitochondrial supernatant into the tubes for a Beckman Ti 35 or Ti 45 rotor, balance the tubes, and ultracentrifuge them at 32,000rpm at 4°C. for 1.6 h (Ti 35) or at 40,000rpm for 1 h (Ti 45) in precooled rotors.
- After centrifugation, discard the clear supernatant and either refill the tubes with postmitochondrial supernatant for the next spin (leave the first pellet in the tube without resuspending it to save time) or remove the microsomal pellets when all the postmitochondrial supernatant has been centrifuged or pellets from three to four ultracentrifugations have been collected in one tube.
- Resuspend the pellets with 1-2 volumes of buffer A supplemented with 0.1 mM PMSF using a 10-ml glass pipette in the reverse orientation (the wide top end down) and then homogenize thoroughly by 10-15 strokes in a Potter-Elvehjem device (overhead drive at slow rotation).
3. Preparation of Crude Clathrim-Coated Vesicles from Microsomal Pellets
Solution
12.5% Ficoll-sucrose: To make 1 liter, dissolve 125g sucrose and 125g Ficoll 400 in buffer A, pH 6.5, and stir overnight in the cold room. Ficoll 400 dissolves only slowly! Keep at 4°C.
Steps
- Mix the well-homogenized microsomes with the same volume of Ficoll-sucrose solution and fill into open tubes suitable for an SS-34 Sorvall rotor. Centrifuge the tubes at 43,000g (19,000rpm) and 4°C for 40 min in a precooled rotor.
- After centrifugation, pour the supernatants containing the clathrin-coated vesicles through a funnel with gauze to remove floating lipids. The compact sediment (about a fifth of the total volume) will stay in the tube and can be discarded.
- Dilute the combined supernatants with 3-4 volumes of cold buffer A containing 0.1 mM PMSE
4. Concentration of Crude Clathrim-Coated Vesicles by Pelleting
Steps
- Ultracentrifuge the coated vesicles in the diluted Ficoll-sucrose solution as described in Section III,C,2. The pellets obtained in this step are considerably smaller than the microsomal pellets. The color usually varies from yellowish-white to brown and is most likely due to contaminating ferritin (Kedersha and Rome, 1986).
- Pour off the supernatants and carefully remove the pellets from the tubes with a spatula. Use a small volume of buffer A + PMSF to wash off material still attached to the walls of the tube.
- Homogenize the combined pellets in about 20 volumes of buffer A + PMSF per bovine brain using first a 10-ml glass pipette in the reverse orientation (the wide top end down) and then homogenize very thoroughly by 10-15 strokes in a Potter-Elvehjem device (overhead drive at slow rotation).
5. Removal of Aggregated Material
Steps
- Fill the crude clathrin-coated vesicles into open tubes for an SS-34 Sorvall rotor. Centrifuge the tubes at 20,000g (13,000rpm) at 4°C for 10min in a precooled rotor.
- After centrifugation, retrieve, pool and place the supernatants on ice. Discard the pellets.
6. Removal of Smooth Membrane Contaminants
Solutions
- Buffer A/D2O: 0.1M MES, 1.0mM EGTA, 0.5mM MgCl2, 0.02% NAN3, pH 6.5. To make 1 liter of 1x buffer A in D2O, dissolve 19.5 g MES, 0.38 g EGTA, and 0.10 g MgCl2 in D2O. Adjust the pH with 5-10 N NaOH solution (prepared in D2O as well) to 6.5, add 2 g NAN3, and bring the volume to 1 liter using D2O. Do not add NAN3 prior to the adjustment of the pH because MES is acidic and may release HN3. Chill to 4°C and supplement with 0.1 mM PMSF prior to use.
- Sucrose/D2O solution: 8% (w/v) sucrose in buffer A/D2O. To make 200 ml, dissolve 16g sucrose in buffer A/D2O and adjust the volume with the same buffer. Keep on ice.
Steps
- Precool an SW 28 rotor and the buckets.
- Pour 30ml of the sucrose/D2O solution per SW 28 centrifugation tube (open top, ultraclear, Beckman #344058). Place the tubes on ice. Carefully overlay 5 ml of the coated vesicles from Section III,C,5 per tube.
- Balance the tubes and assemble the buckets and the rotor. Spin the rotor in an ultracentrifuge at 110,000g (25,000rpm) at 4°C for 2h.
- Remove the supernatants and resuspend the pellets in 5-10 volumes of buffer A plus PMSF using a pipette and a suitable homogenizer.
- Material obtained after this step can either be used directly or be frozen in liquid nitrogen and stored at -80°C.
IV. COMMENTS
Following this protocol (for summary, see Fig. 1), near homogeneous preparations of clathrin-coated vesicles are obtained from bovine brain. The yields usually are about 50-100mg clathrin-coated vesicles/ kg brain cortex.
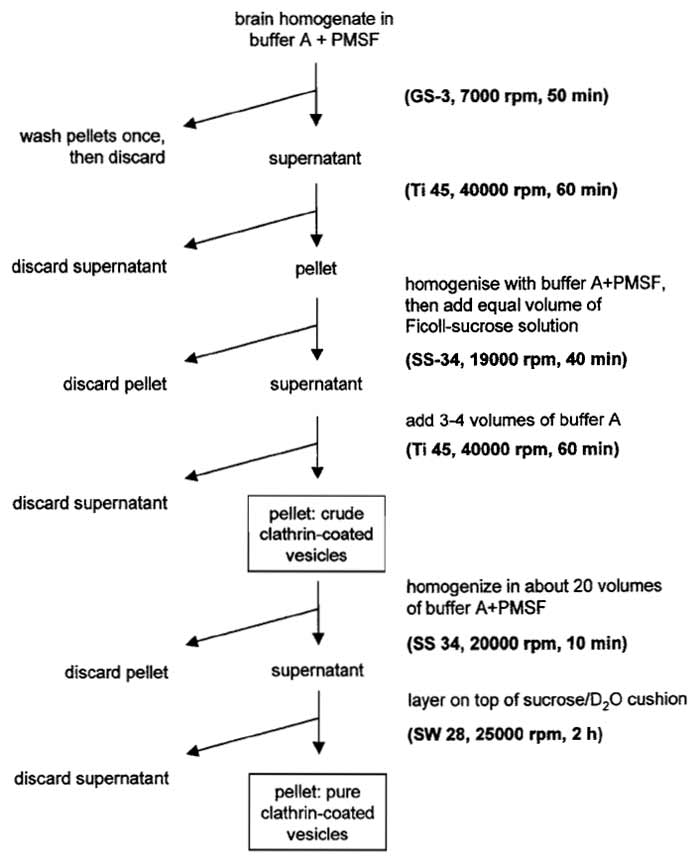 |
| FIGURE 1 Purification scheme for clathrin-coated vesicles. |
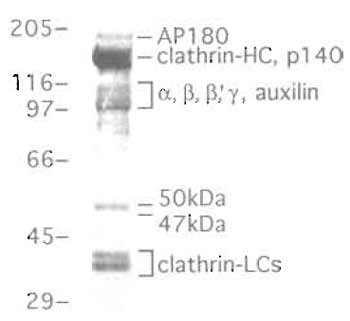 |
| FIGURE 2 Coat proteins obtained by Tris extraction of a crude clathrin-coated vesicle preparation from bovine brain as described in the text. Coated vesicles were incubated in 0.5M Tris, pH 7.0, to release the peripheral membrane proteins and were then ultracentrifuged to remove the membranes. Approximately 12µg Tris extract was electrophoresed in a low-Bis SDS-polyacrylamide gel (for details, see Lindner and Ungewickell, 1992). Note that in this gel system the brain-specific, clathrin- associated protein AP180 is well resolved from the clathrin heavy chain. Both AP180 and auxilin are only detectable in brain-derived, clathrin-coated vesicles. In nonneuronal tissues, homologues are expressed (CALM for AP180 and GAK/auxilin2 for auxilin). |
This basic protocol can also be used with other organs rich in clathrin-coated membranes, such as adrenal glands, liver, or placenta. For adrenal glands, a more thorough homogenization (usually 6 or more 10-s bursts in a Waring commercial blender) is required to break up the very resistant capsule material.
Clathrin-coated vesicle preparations from liver usually contain a considerable amount of ribonucleoprotein complexes termed vaults. It is advisable to remove these structures by velocity centrifugation on 5-40% sucrose gradients as an additional step after the preparation described earlier (for details, see Kedersha and Rome, 1986). Vaults are at least partially dissociated by the conventional extraction methods for clathrin coat structures and thus contaminate coat protein extracts.
For the preparation of fusion-competent, clathrincoated vesicle from cell culture cells, a more elaborate protocol has been described (Woodman and Warren, 1991)
V. PITFALLS
Do not mix the Ficoll-sucrose solution to microsomal pellets obtained in Section III,C,3 before thorough homogenization of the pellets. The high viscosity of the Ficoll-sucrose solution prevents proper homogenization.
In order to quantitatively pellet the clathrin-coated vesicles from the supernatant after centrifugation in Ficoll-sucrose, dilute with a minimum of 3 volumes of buffer A + PMSF to decrease both the density and the viscosity of the supernatant.
References
Ahle, S., Mann, A., Eichelsbacher, U., and Ungewickell, E. (1988). Structural relationships between clathrin assembly proteins from the Golgi and the plasma membrane. EMBO J. 7, 919-929.
Ahle, S., and Ungewickell, E. (1990). Auxilin, a newly identified clathrin-associated protein in coated vesicles from bovine brain. J. Cell Biol. 111, 19-29.
Bonifacino, J. S., and Lippincott-Schwartz, J. (2003). Coat proteins: Shaping membrane transport. Nature Rev. Mol. Cell Biol. 4, 409-414.
Brodsky, E M., Chen, C. Y., Knuehl, C., Towler, M. C., and Wakeham, D. E. (2001). Biological basket weaving: Formation and function of clathrin-coated vesicles. Annu. Rev. Cell Dev. Biol. 17, 517-568.
Campbell, C., Squicciarini, J., Shia, M., Pilch, R E, and Fine, R. E. (1984). Identification of a protein kinase as an intrinsic component of rat liver coated vesicles. Biochemistry 23, 4420-4426.
De Camilli, P., Chen, H., Hyman, J., Panepucci, E., Bateman, A., and Brunger, A. T. (2002). The ENTH domain. FEBS Lett. 513, 11-18.
Kanaseki, T., and Kadota, K. (1969). The "vesicle in a basket". A morphological study of the coated vesicle isolated from the nerve endings of the guinea pig brain, with special reference to the mechanism of membrane movements. J. Cell Biol. 42, 202-220.
Kalthoff, C., Alves, J., Urbanke, C., Knorr, R., and Ungewickell, E. J. (2002). Unusual structural organization of the endocytic proteins AP180 and epsin 1. J. Biol. Chem. 277, 8209-8216.
Kedersha, N. L., and Rome, L. H. (1986). Isolation and characterization of a novel ribonucleoprotein particle: Large structures contain a single species of small RNA. J. Cell Biol. 103, 699- 709.
Kirchhausen, T. (1999). Adaptors for clathrin-mediated traffic. Annu. Rev. Cell Dev. Biol. 15, 705-732.
Lindner, R., and Ungewickell, E. (1991). Light-chain-independent binding of adaptors, AP180 and auxilin to clathrin. Biochemistry 30, 9097-9101.
Lindner, R., and Ungewickell, E. (1992). Clathrin-associated proteins from bovine brain coated vesicles: An analysis of their number and assembly-promoting activity. J. Biol. Chem. 267, 16567-16573.
Maycox, P. R., Link, E., Reetz, A., Morris, S. A., and Jahn, R. (1992). Clathrin-coated vesicles in nervous tissue are involved primarily in synaptic vesicle recycling. J. Cell Biol. 118, 1379-1388.
Raiborg, C., Bache, K. G., Gillooly, D. J., Madshus, I. H., Stang, E., and Stenmark, H. (2002). Hrs sorts ubiquitinated proteins into clathrin-coated microdomains of early endosomes. Nature Cell Biol. 4, 394-398.
Sachse, M., Urbe, S., Oorschot, V., Strous, G. J., and Klumperman, J. (2002). Bilayered clathrin coats on endosomal vacuoles are involved in protein sorting toward lysosomes. Mol. Biol. Cell 13, 1313-1328.
Schmid, S. L. (1997). Clathrin-coated vesicle formation and protein sorting: An integrated process. Annu. Rev. Biochem. 66, 511-548.
Stoorvogel, W., Oorschot, V., and Geuze, H. J. (1996). A novel class of clathrin-coated vesicles budding from endosomes. J. Cell Biol. 132, 21-33.
Traub, L. M., Bannykh, S. I., Rodel, J. E., Aridor, M., Balch, W. E., and Kornfeld, S. (1996). AP-2-containing clathrin coats assemble on mature lysosomes. J. Cell Biol. 135, 1801-1814.
Ungewickell, E., Ungewickell, H., Holstein, E. H., Lindner, R., Prasad, K., Barouch, W., Martin, B., Greene, L. E., and Eisenberg, E. (1995). Role of auxilin in uncoating clathrin-coated vesicles. Nature 378, 632-635.
Wasiak, S., Legendre-Guillemin, V., Puertollano, R., Blondeau, E, Girard, M., de Heuvel, E., Boismenu, D., Bell, A. W., Bonifacino, J. S., and McPherson, P. S. (2002). Enthoprotin: A novel clathrinassociated protein identified through subcellular proteomics. J. Cell Biol. 158, 855-862.
Woodman, P. G., and Warren, G. (1991). Isolation and characterization of functional clathrin-coated vesicles. J. Cell Biol. 112, 1133-1141.




