Action of Enzymes
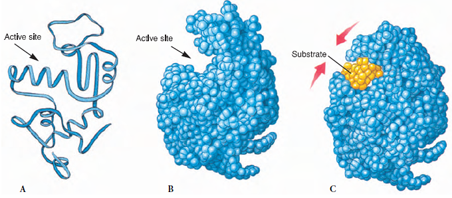
Figure 4-4 How an enzyme works. This space-filling model shows that the enzyme lysozyme bears a pocket containing the active site. When a chain of sugars (substrate) enters the pocket, the protein enzyme changes shape slightly so that the pocket enfolds the substrate and conforms to its shape. This positions the active site (an amino acid in the protein) next to a bond between adjacent sugars in the chain, causing the sugar chain to break.
If the formation of an enzyme-substrate complex is so rapidly followed by dissociation, how can biochemists be certain that an ES complex exists? The original evidence offered by Leonor Michaelis in 1913 is that, when the substrate concentration is increased while the enzyme concentration is held constant, the reaction rate reaches a maximum velocity.This saturation effect is interpreted to mean that all catalytic sites become filled at high substrate concentration. It is not seen in uncatalyzed reactions. Other evidence includes the observation that the ES complex displays unique spectroscopic characteristics not displayed by either the enzyme or the substrate alone. Furthermore, some ES complexes can be isolated in pure form, and at least one kind (nucleic acids and their polymerase enzymes) has been directly visualized with the electron microscope.

Figure 4-4 How an enzyme works. This space-filling model shows that the enzyme lysozyme bears a pocket containing the active site. When a chain of sugars (substrate) enters the pocket, the protein enzyme changes shape slightly so that the pocket enfolds the substrate and conforms to its shape. This positions the active site (an amino acid in the protein) next to a bond between adjacent sugars in the chain, causing the sugar chain to break.
Enzymes that engage in important main-line sequences—such as the crucial energy-providing reactions of the cell that proceed constantly—seem to operate in sets rather than in isolation. For example, conversion of glucose to carbon dioxide and water proceeds through 19 reactions, each requiring a specific enzyme. Main-line enzymes are found in relatively high concentrations in the cell, and they may implement quite complex and highly integrated enzymatic sequences. One enzyme carries out the first step, then passes the product to another enzyme that catalyzes another step, this process continuing until the end of the enzymatic pathway is reached. The reactions may be said to be coupled. Coupled reactions will be explained in a following section on chemical energy transfer by ATP.




