Enzyme-Catalyzed Reactions
Enzyme-Catalyzed
Reactions
Enzyme-catalyzed reactions are reversible, which is signified by the double arrows between substrate and products. For example:
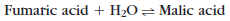
However, for various reasons the reactions catalyzed by most enzymes tend to go predominantly in one direction. For example, the proteolytic enzyme pepsin degrades proteins into amino acids (a catabolic reaction), but it does not accelerate the rebuilding of amino acids into any significant amount of protein (an anabolic reaction). The same is true of most enzymes that catalyze the cleavage of large molecules such as nucleic acids, polysaccharides, lipids, and proteins. There is usually one set of reactions and enzymes that break them down (catabolism; Gr. kata, down, + bole, throw), but they must be resynthesized by a different set of reactions that are catalyzed by different enzymes (anabolism; Gr. ana, up, + bole, throw). This apparent irreversibility exists because the chemical equilibrium usually favors the formation of the smaller degradation products.
The net direction of any chemical reaction depends on the relative energy contents of the substances involved. If there is little change in the chemical bond energy of the substrate and the products, the reaction is more easily reversible. However, if large quantities of energy are released as the reaction proceeds in one direction, more energy must be provided in some way to drive the reaction in the reverse direction. For this reason many if not most enzyme-catalyzed reactions are in practice irreversible unless the reaction is coupled to another that makes energy available. In the cell both reversible and irreversible reactions are combined in complex ways to make possible both synthesis and degradation.
Hydrolysis literally means “breaking with water.” In hydrolysis reactions, a molecule is broken down by the addition of water.A hydrogen is attached to one subunit and a hydroxyl (−OH) unit is attached to another. This breaks the covalent bond between subunits. Hydrolysis is the opposite of condensation (water-losing) reactions in which the subunits of molecules are linked together by the removal of water. Macromolecules are built by condensation reactions.
Enzyme-catalyzed reactions are reversible, which is signified by the double arrows between substrate and products. For example:
However, for various reasons the reactions catalyzed by most enzymes tend to go predominantly in one direction. For example, the proteolytic enzyme pepsin degrades proteins into amino acids (a catabolic reaction), but it does not accelerate the rebuilding of amino acids into any significant amount of protein (an anabolic reaction). The same is true of most enzymes that catalyze the cleavage of large molecules such as nucleic acids, polysaccharides, lipids, and proteins. There is usually one set of reactions and enzymes that break them down (catabolism; Gr. kata, down, + bole, throw), but they must be resynthesized by a different set of reactions that are catalyzed by different enzymes (anabolism; Gr. ana, up, + bole, throw). This apparent irreversibility exists because the chemical equilibrium usually favors the formation of the smaller degradation products.
The net direction of any chemical reaction depends on the relative energy contents of the substances involved. If there is little change in the chemical bond energy of the substrate and the products, the reaction is more easily reversible. However, if large quantities of energy are released as the reaction proceeds in one direction, more energy must be provided in some way to drive the reaction in the reverse direction. For this reason many if not most enzyme-catalyzed reactions are in practice irreversible unless the reaction is coupled to another that makes energy available. In the cell both reversible and irreversible reactions are combined in complex ways to make possible both synthesis and degradation.
Hydrolysis literally means “breaking with water.” In hydrolysis reactions, a molecule is broken down by the addition of water.A hydrogen is attached to one subunit and a hydroxyl (−OH) unit is attached to another. This breaks the covalent bond between subunits. Hydrolysis is the opposite of condensation (water-losing) reactions in which the subunits of molecules are linked together by the removal of water. Macromolecules are built by condensation reactions.




