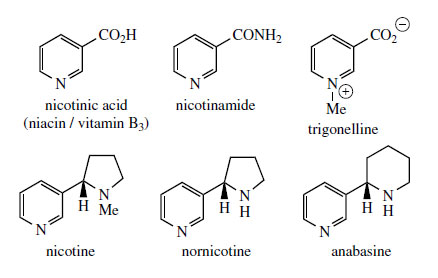
Pyridine Alkaloids
The alkaloids found in tobacco* (
Nicotiana tabacum; Solanaceae) include
nicotine and
anabasine (Figure 28). The structures contain a pyridine ring together with a pyrrolidine ring (in nicotine) or a piperidine unit (in anabasine), the latter rings arising from ornithine and lysine respectively. The pyridine unit has its origins in
nicotinic acid (vitamin B3)* (Figure 28), the vitamin sometimes called
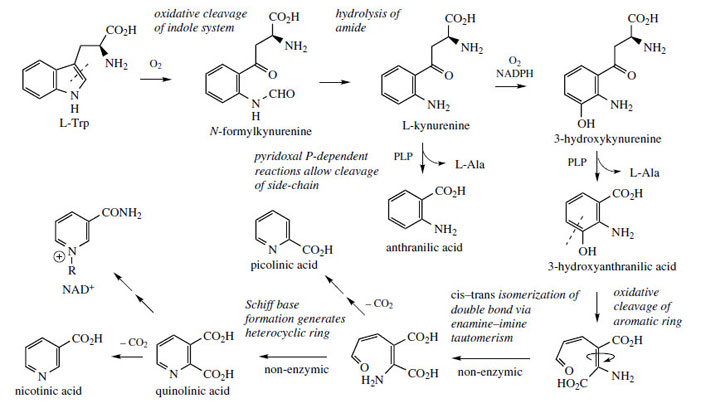
niacin, which forms an essential component of coenzymes such as NAD+ and NADP+. The nicotinic acid component of nicotinamide is synthesized in animals by degradation of L-tryptophan through the
kynurenine pathway and
3-hydroxyanthranilic acid (Figure 29), the pyridine ring being formed by oxidative cleavage of the benzene ring and subsequent inclusion of the amine nitrogen (Figure 29). However, plants such as
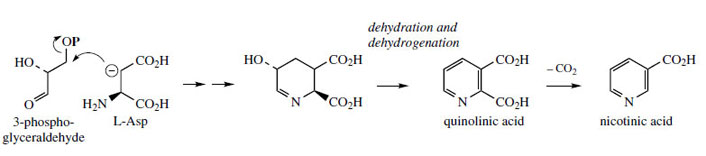
Nicotiana use a different pathway employing glyceraldehyde 3-phosphate and Laspartic acid precursors (Figure 30). The dibasic acid
quinolinic acid features in both pathways, decarboxylation yielding nicotinic acid.
 |
| Figure 28 |
 |
| Figure 29 |
 |
| Figure 30 |
Vitamin B3
Vitamin B3 (nicotinic acid, niacin) (Figure 28) is a stable water-soluble vitamin widely distributed in foodstuffs, especially meat, liver, fish, wheat germ, and yeast. However, in some foods, e.g. maize, it may be present in a bound form, and is not readily available. Diets based principally on maize may lead to deficiencies. The amino acid tryptophan can be converted in the body into nicotinic acid (Figure 29), and may provide a large proportion of the requirements. Nicotinic acid is also produced during the roasting of coffee from the decomposition of the N-methyl derivative trigonelline (Figure 28). Nicotinic acid is converted into nicotinamide (Figure 28), though this compound also occurs naturally in many foods. The term vitamin B3 is often used for the combined nicotinamide–nicotinic acid complement. In the form of the coenzymes NAD+ and NADP+, nicotinamide plays a vital role in oxidation–reduction reactions, and is the most important electron carrier in primary metabolism. Deficiency in nicotinamide leads to pellagra, which manifests itself in diarrhoea, dermatitis, and dementia. Oral lesions and a red tongue may be more noticeable than the other symptoms. Nicotinamide is usually preferred over nicotinic acid for dietary supplements since there is less risk of gastric irritation. Both are produced synthetically. It is common practice to enrich many foods, including bread, flour, corn, and rice products.
Nicotinic acid in large doses can lower both cholesterol and triglyceride concentrations by inhibiting their synthesis.
 |
| Figure 31 |
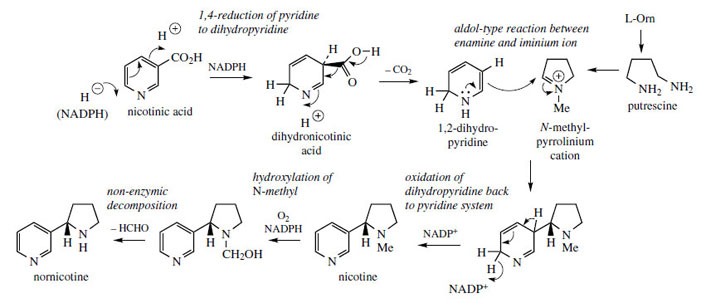
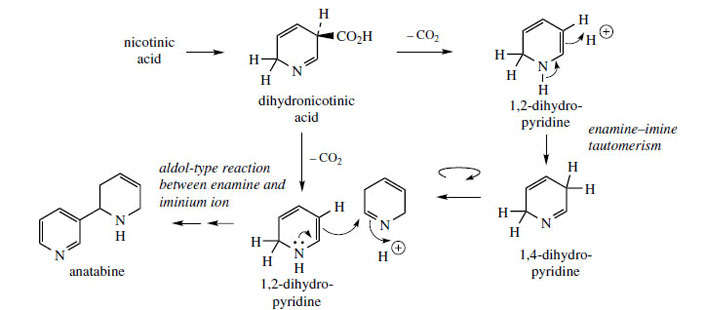
In the formation of
nicotine, a pyrrolidine ring derived from ornithine, most likely as the N-methyl-Δ
1-pyrrolinium cation (Figure 2) is attached to the pyridine ring of nicotinic acid, displacing the carboxyl during the sequence (Figure 31). A dihydronicotinic acid intermediate is likely to be involved allowing decarboxylation to the enamine 1,2-dihydropyridine. This allows an aldol-type reaction with the N-methylpyrrolinium cation, and finally dehydrogenation of the dihydropyridine ring back to a pyridine gives nicotine.
Nornicotine is derived by oxidative demethylation of nicotine.
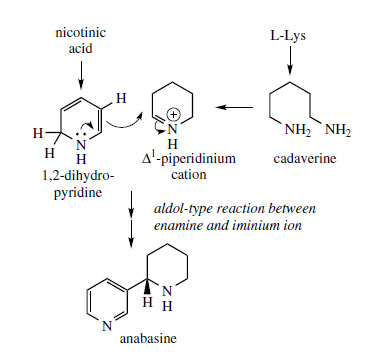
Anabasine is produced from nicotinic acid and lysine via the Δ
1-piperidinium cation in an essentially analogous manner (Figure 32). A subtle anomaly has been exposed in that a further
Nicotiana alkaloid
anatabine appears to be derived by combination of two nicotinic acid units, and the Δ
3-piperideine ring is
not supplied by lysine (Figure 33).
 |
| Figure 32 |
Nicotinic acid undoubtedly provides the basic skeleton for some other alkaloids.
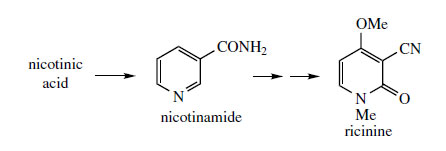
Ricinine (Figure 35) is a 2-pyridone structure and contains a nitrile grouping, probably formed by dehydration of a nicotinamide derivative. This alkaloid is a toxic constituent of castor oil seeds (
Ricinus communis; Euphorbiaceae), though the toxicity of the seeds results mainly from the polypeptide ricin.
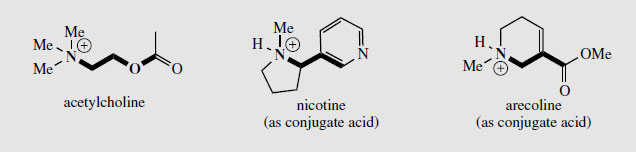
Arecoline (Figure 36) is found in Betel nuts (
Areca catechu: Palmae/Arecaceae)* and is a tetrahydronicotinic acid derivative. Betel nuts are chewed in India and Asia for the stimulant effect of arecoline.
 |
| Figure 33 |
Tobacco
Tobacco is the cured and dried leaves of
Nicotiana tabacum (Solanaceae), an annual herb
indigenous to tropical America, but cultivated widely for smoking. Tobacco leaves may contain
from 0.6–9% of (-)-nicotine (Figure 28), an oily, volatile liquid alkaloid, together with smaller
amounts of structurally related alkaloids, e.g. anabasine and nornicotine (Figure 28). In
the leaf, the alkaloids are typically present as salts with malic and citric acids. Nicotine in
small doses can act as a respiratory stimulant, though in larger doses it causes respiratory
depression. Despite the vast array of evidence linking tobacco smoking and cancer, the
smoking habit continues throughout the world, and tobacco remains a major crop plant.
Tobacco smoke contains a number of highly carcinogenic chemicals formed by incomplete
combustion, including benzpyrene, 2-naphthylamine, and 4-aminobiphenyl. Metabolism by
the body’s P-450 system leads to further reactive intermediates, which can combine with
DNA and cause mutations. Tobacco smoking also contributes to atherosclerosis, chronic bronchitis, and emphysema, and is regarded as the single most preventable cause of death
in modern society.
Nicotine is being used by former smokers who wish to stop the habit. It is
available in the form of chewing gum or nasal sprays, or can be absorbed transdermally from
nicotine-impregnated patches.
 |
| Figure 34 |
Powdered tobacco leaves have long been used as an insecticide, and nicotine from
Nicotiana tabacum or N. rustica has been formulated for agricultural and horticultural use. The
free base is considerably more toxic than salts, and soaps were included in the formulations
to ensure a basic pH and to provide a surfactant. Other
Nicotiana alkaloids, e.g. anabasine
and nornicotine, share this insecticidal activity. Although an effective insecticide, nicotine has
been replaced by other agents considered to be safer. Nicotine is toxic to man due to its
effect on the nervous system, interacting with the nicotinic acetylcholine receptors, though
the tight binding observed is only partially accounted for by the structural similarity between
acetylcholine and nicotine (Figure 34). Recent studies suggest that nicotine can improve
memory by stimulating the transmission of nerve impulses, and this finding may account
for the lower incidence of Alzheimer’s disease in smokers. Any health benefits conferred
by smoking are more than outweighed by the increased risk of heart, lung, and respiratory
diseases.
 |
| Figure 35 |












