Enzyme Immunoassay (Eia)
As in fluorescent antibody tests, the antibody in EIAs is conjugated with a marker that can be detected when an antigen-antibody reaction has taken place. In EIAs, the marker is an enzyme, typically alkaline phosphatase or horseradish peroxidase. These enzymes catalyze the breakdown of a colorless substrate to a colored end-product. To visualize the binding of antigen and the enzyme-linked antibody, the appropriate substrate for the enzyme must be added. A positive reaction results in the production of a colored end-product that can be detected visually or measured quantitatively in a spectrophotometer.Two basic formats for EIA testing are in use. In the first, monoclonal antibody specific for the antigen sought is bound to a solid surface such as plastic tubes, beads, or wells of a microtiter tray. The clinical sample is added to this solid surface followed by incubation and washing steps. If the antigen is present in the sample, it will bind to the antibody and unbound material is washed away. Now, the enzyme-labeled antibody is added to detect the antigen-antibody complex. This step is accomplished either in a direct or indirect manner. In the direct method, the second antibody, which is conjugated to the enzyme, reacts with antigen bound by the first antibody on the solid surface. In the indirect method, two additional antibodies are needed to develop the reaction. The first is unlabeled antibody specific for the bound antigen and the second is an enzyme-labeled antibody that reacts with the first antibody. In this way, the indirect EIA is similar to the IFA test. Finally, in both test methods the substrate for the enzyme is added. The amount of colored end-product that develops indicates the amount of antigen present in the clinical sample. Figure 19.3 illustrates direct and indirect EIA methods.
EIA kits for detecting certain parasites, rotavirus, and other enteric viruses, and toxins of diarrhea pathogens, are available in this format. Colorplate 25 illustrates such a kit.
|
|||
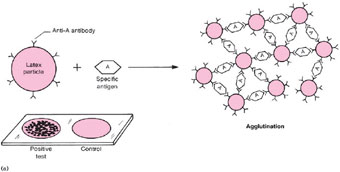
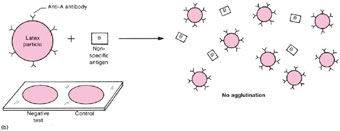
| Figure 19.2 Diagram of a latex reaction. (a) When latex particles that are coated with an antibody (for example, group A Streptococcus antibody) react with a specific antigen (group A Streptococcus antigen), the particles join together to form clumps that agglutinate on the test slide (lower left corner of (a)). In the control test that is always run in parallel on the same slide, the same antigen is mixed with latex particles that are not coated with antibody, therefore, the particles remain in suspension and do not agglutinate. In diagram section (b), the antibody-coated particles do not react with the nonspecific antigen (for example, group B Streptococcus antigen), therefore, no clumps are formed, and the test as well as the control suspension shows no agglutination (lower left corner of (b)). |
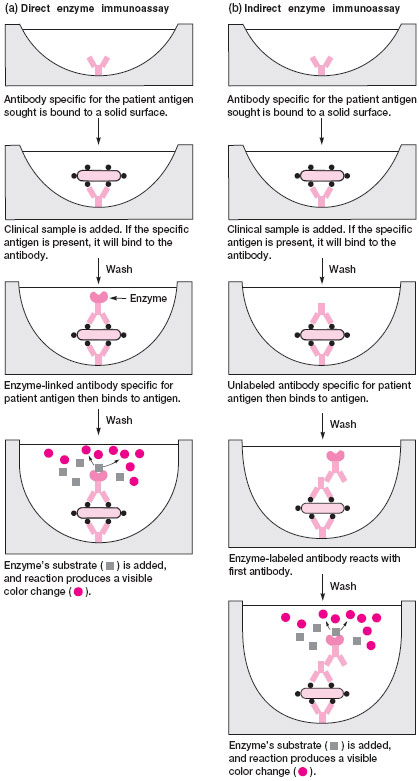 |
| Figure 19.3 Direct and indirect enzyme immunoassays. |
In another format for performing EIA tests, the specific enzyme-labeled antibody is bound to a porous nitrocellulose or nylon membrane. The membrane is contained in a disposable plastic cassette with a small chamber to which the liquid clinical sample can be added (fig. 19.4). An absorbent material on the underside of the membrane serves to draw the liquid sample through the membrane. If antigen is present in the sample, it will bind to the membrane-bound antibody as the material passes through the membrane. After the colorless substrate is added and passed through the membrane filter, development of a colored area indicates a positive test (fig. 19.4).
Membrane-bound EIA tests are very popular because they are easy to perform and reliable results are usually available within 5 to 10 minutes. They are available for direct specimen detection of some bacteria and viruses.
 |
| Figure 19.4 An enzyme immunoassay kit for “strep” throat. The left side illustrates a negative test, with only the control (C) line positive. On the right, both the control (C) and patient specimen (T) are positive as illustrated by the appearance of two lines. |