Fungi: Yeasts and Molds
Medical mycology is concerned with the study and identification of the pathogenic yeasts and molds, collectively called fungi (sing., fungus). You should be familiar with a number of important mycotic diseases.Yeasts are unicellular fungi that reproduce by budding, that is, by forming and pinching off daughter cells (see colorplate 41). Yeast cells are much larger (about five to eight times) than bacterial cells. The best-known (and most useful) species is “bakers’ yeast,” Saccharomyces cerevisiae, used in bread making and in fermentations for wine and beer production.
Molds are multicellular, higher forms of fungi. They are composed of filaments called hyphae, abundantly interwoven in a mat called the mycelium. Specialized structures for reproduction arise from the hyphae and produce conidia (also called spores), each of which can germinate to form new growth of the fungus. The visible growth of a mold often has a fuzzy appearance because the mycelium extends upward from its vegetative base of growth, thrusting specialized hyphae that bear conidia into the air. This portion is called the aerial mycelium. You have often seen this on moldy bread or other food, and you have probably also noted that different molds vary in color (black, green, yellow) because of their conidial pigment (see colorplate 42).
Most of the thousands of species of yeasts and molds that are found in nature are saprophytic and incapable of causing disease. Indeed, many are extremely useful in the processing of certain foods (such as cheeses) and as a source of antimicrobial agents. Penicillium notatum, for example, is the mold that produces penicillin.
Mycotic Diseases and Their Agents
Fungal diseases fall into four clinical patterns: superficial infections on surface epithelial structures (skin, hair, nails), systemic infections of deep tissues, and subcutaneous and opportunistic infections.
Superficial Mycoses
The pathogenic fungi that cause infections of skin, hair, or nails are often referred to collectively as dermatophytes. There are three major genera of dermatophytes:
- Trichophyton:
This genus contains many species (e.g., T. mentagrophytes,T. rubrum,T. tonsurans) associated with “ringworm”infections of the scalp, body, nails, and feet. “Athlete’s foot” is perhaps the most common of these infections. - Microsporum:
There are three common species of this genus: M. audouini, M. canis, and M. gypseum. These fungi cause ringworm infections of the hair and scalp, and also of the body. - Epidermophyton:
One species, E. floccosum, causes ringworm of the body, including “athlete’s foot.” It does not affect hair or nails.
Systemic and Subcutaneous Mycoses
Many of the fungi involved in systemic and subcutaneous infections are either yeasts or display both a yeast and a mold phase (they are said to be dimorphic because of this). The yeast phase of dimorphic fungi grows best at 35 to 37°C, whereas their mold phase grows optimally at a lower (25°C) temperature. The most important pathogenic fungi that cause systemic or subcutaneous disease are shown in table 31.1.
Opportunistic Mycoses
Under ordinary circumstances, fungi are of low pathogenicity and have little ability to invade the human body. However, when the host’s immune defense mechanisms are decreased by illness (leukemias, lymphomas, acquired immunodeficiency syndrome) or by drugs (steroids, cancer chemotherapeutics, transplantation drugs), fungi (as well as other microorganisms) find the opportunity to invade and establish disease. Because few antimicrobial agents are available to combat fungal infections, these represent among the most serious opportunistic illnesses and frequently are the direct cause of the patient’s death. Some opportunistic fungi, such as the yeasts Candida and Cryptococcus (see colorplates 41 and 43), are not always associated with immunosuppression, but others, especially species of Aspergillus (see colorplates 44 and 45) and Mucor, infect only “disabled” hosts. Because the latter organisms are also widespread in the environment, health care personnel must be certain that specimens obtained from immunocompromised patients are always collected in sterile containers and in such a manner as to avoid contamination with airborne fungal conidia. The microbiology technologist must also protect culture plates and broths from such contamination so that any molds that grow out are known to come from the patient and not the environment. Some agents of opportunistic fungal infections are listed in table 31.1.
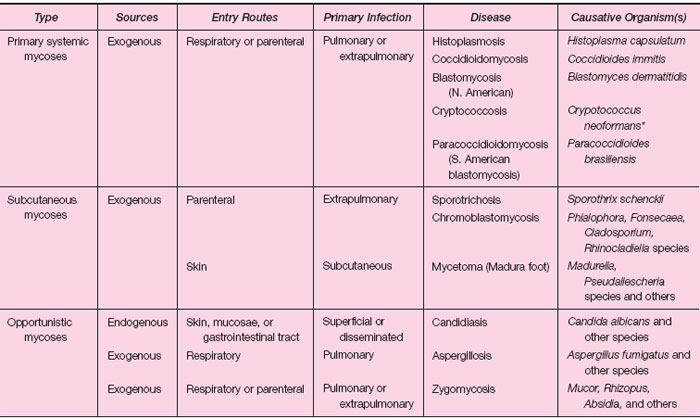 |
| Table 31.1 Classification of Systemic and Subcutaneous Mycoses |
Laboratory Diagnosis
The laboratory diagnosis of a fungal infection depends on the direct microscopic detection of fungal structures in clinical samples and/or the recovery in culture and subsequent identification of the fungus. Fungi may be isolated from a variety of clinical specimens representing the focus of infection (sputum, spinal fluid, tissue, pus aspirated from lymph nodes or other lesion, bone marrow aspirates, skin scrapings). All specimens of sufficient quantity submitted for fungal culture should be examined microscopically for fungi. When there is not sufficient specimen to allow both a culture and direct microscopic examination, the culture has priority over the smear because culture is more sensitive than microscopic examination. However, observing a fungus in a clinical specimen is often valuable in establishing the significance of the fungus (i.e., ruling out contamination) and in providing early information that may be crucial for determining appropriate patient therapy.
In general, serological tests (looking for a significant change in antibody titer in paired serum specimens, have limited application for the diagnosis of most fungal infections. Exceptions to this rule include certain dimorphic fungal diseases, such as histoplasmosis and coccidioidomycosis. The purpose of this laboratory exercise is to acquaint the student with some direct microscopic and cultural methods that are available for establishing the laboratory diagnosis of a human mycosis.
Direct Microscopic Examinations
- Histopathology:
The visualization of fungal structures (hyphae, conidia, etc.) in tissue obtained by biopsy or at autopsy establishes the involvement of the fungus in human disease. Specialized tissue stains such as Giemsa, methenamine silver (see colorplate 45), or mucicarmine may be used to facilitate the detection of the fungus in tissue. The particular fungal structures that are seen in tissue can sometimes confirm the identity of the fungus (e.g., spherules [the yeast form] of Coccidioides immitis [see fig. 31.1] or cysts of Pneumocystis carinii [see colorplate 46 ]) or suggest the presence of a particular fungal group. In this latter case, culture is used to confirm the presence and identity of the fungal pathogen. - Direct Smears:
Direct smears of patient material other than tissue are often made to detect the presence of fungal elements microscopically. Several types of stains or reagents are used to facilitate the detection of certain fungi.- Ten percent potassium hydroxide: Potassium hydroxide preparations are used to examine a variety of clinical samples including hair, nails, skin scrapings, fluids, or exudates. The potassium hydroxide solution serves to clear away tissue cells
- and debris, making the fungi more prominent. Slides must be examined with reduced illumination to allow fungal structures to be seen (see colorplate 47).
- Calcofluor white:This reagent is used with most specimen types to detect the presence of fungi by fluorescence microscopy. The cell walls of the fungi bind the stain and fluoresce blue-white or apple green, depending on the filter combination used with the microscope. This stain is useful for examining skin scrapings for the presence of dermatophytes and tissues and body fluids for yeast and filamentous fungi (see colorplate 45).
- India ink:This traditional test is usually ordered to screen for the presence of Cryptococcus neoformans in spinal fluid samples. This yeast is encapsulated, and the capsule can be visualized readily against the black background of the India ink as a clear halo surrounding the yeast cell (see colorplate 43). The India ink test is very insensitive (detecting only 40% of cases of cryptococcal meningitis) and therefore has been superseded by other tests, such as the cryptococcal antigen latex agglutination test, which detects more than 90% of cases of cryptococcal meningitis. The India ink test is rarely performed in clinical microbiology laboratories.
- Wright, Giemsa, or Diff-Quik stains. These specialized stains are often used on blood and bone marrow smears to look for intracellular yeast forms of Histoplasma capsulatum.
- Gram stain: Most fungi are not stained well by the Gram-stain procedure, and therefore, it is of limited use when
examining specimens for fungal forms. It is generally reliable only for detecting the presence of Candida species (see colorplate 41), Sporothrix schenkii, and perhaps a few other fungi in clinical material. In Gram-stained spinal fluid
specimens, Cryptococcus neoformans may appear as irregularly staining gram-positive yeast cells surrounded by an orange
capsule(see colorplate 43).
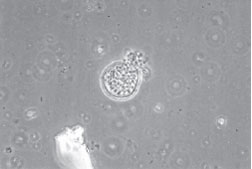
Figure 31.1 KOH preparation of lung biopsy material showing spherule (yeast form) of Coccidioides immitis. Many endospores bud off from the thick-walled spherule, which has burst, releasing the endospores into the surrounding tissue. Each endospore is able to form a new spherule. In culture, this dimorphic fungus will grow as the filamentous hyphal form.
- Culture:
The cultural isolation of a fungus from a clinical specimen and its subsequent identification is the definitive test for establishing the etiology of a fungal disease. The medium most commonly used to isolate fungi from clinical specimens is Sabouraud dextrose agar. Most fungi grow well at room temperature; however, depending on the fungus, several days to weeks or months may be required for its recovery and complete characterization. The following discussion summarizes some of the cultural procedures used to identify yeasts and molds. - Yeasts:
Yeasts, such as Candida species and Cryptococcus neoformans, are a heterogeneous group. Their identification is based on colonial and cellular morphology and biochemical characteristics. Morphology is used primarily to establish the genus identification, whereas biochemical tests are used to differentiate the various species. - Germ tube test: More than 90% of yeast infections are caused by Candida albicans. The germ tube test is a rapid and inexpensive method used to identify this species. When they are inoculated into a tube containing 1 ml of horse serum, all strains of C. albicans produce a specialized structure, called a germ tube, within 2 hours of incubation at 35°C (see fig. 31.2). All other yeast isolates are “germ tube negative” within that same time period, but prolonged incubation past 2 hours may result in false-positive tests.
- Biochemical characterization:Traditional tests used for identifying yeasts to the species level involve the assimilation and/or degradation of various carbohydrates. These tests are now commercially available in kit form, much as bacterial identification systems are (see Experiment 24.6). Popular systems include the Minitek, the API 20C Yeast Identification System, the Uni-Yeast-Tek System, and the automated bioMerieux-Vitek YBC, all of which are modifications of the classic carbohydrate degradation and assimilation techniques. Identification results are usually available within 24 to 72 hours.
- Molds:
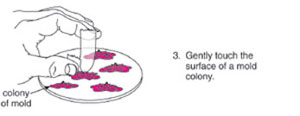
The identification of filamentous fungi (molds) depends on a number of factors including growth rate, colonial appearance, microscopic morphology, and some metabolic properties. A highly experienced technologist or mycologist is needed to identify most molds reliably.- Macroscopic appearance:A giant colony culture (a single colony grown on the center of a culture plate) is often prepared to determine the growth rate of a mold and to observe its colonial appearance (color, texture of hyphae, etc.) (see colorplate 42). The bottom side of the plate (called the “reverse”) is also examined because some fungi produce a diffusible pigment that is evident from the reverse side only. These macroscopic features are useful in the preliminary identification of the fungus.
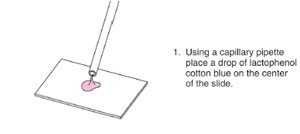
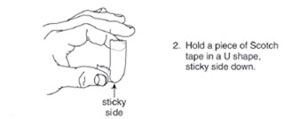
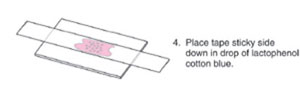
- Microscopic appearance:Accurate identification of a mold is based on microscopic examination of the conidia and the fungal structures on which they are borne. Microscopic preparations may be made directly from the culture (see colorplate 48), or mycologists may use a slide culture technique that allows these sporulating structures to be viewed microscopically at various stages of growth without disturbing their characteristic arrangements. To prepare a slide culture, a small square block of Sabouraud agar is placed on a sterile microscope slide in a sterile petri dish. The agar is inoculated with the fungus to be identified and then covered with a cover glass. A piece of wet cotton is placed in the dish to keep the atmosphere moist and prevent drying of the agar medium. The dish and slide are incubated at room temperature or in a 25 to 30°C incubator. The slide can be viewed directly under the microscope, or the cover glass can be removed, stained with lactophenol cotton blue, and mounted on a clean slide for viewing (see colorplate 44). This slide culture system improves the chances of observing fungal structures that permit genus and species identification.
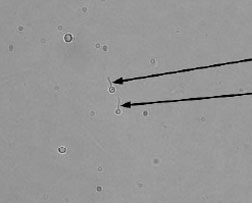
Figure 31.2 Germ-tube formation by Candida albicans. The two yeast cells in the center have sprouted a germ tube (arrows) when incubated for 2 hours in horse serum. Not all cells in the preparation will form the germ tube. The halo around the cells represents light refraction and not a capsule.
| Purpose | To observe the microscopic structures of some fungi |
| Materials | Sabouraud agar slant culture of Candida albicans Tubes containing 1.0 ml of inactivated horse serum Sabouraud agar plate cultures of Aspergillus, Rhizopus, Penicillium Blood agar plates exposed 3 to 5 days earlier for 30 minutes at home, in class, public transportation, etc. Glass microscope slides and coverslips Transparent tape (e.g., Scotch tape) Dropper bottles containing lactophenol cotton blue and methylene blue Capillary pipettes and pipette bulbs Prepared slides of dermatophytes Prepared slides of yeast and mold phases of a systemic fungus Projection slides if available |
Procedures
|
|
 |
| Table 31.2 Some Important Pathogenic Fungi |
Results
- Draw a diagram showing all the structures of Candida albicans that you observed.
- List the principal differences you have observed in yeast cells as compared with bacteria.
- Draw the conidia, conidia-bearing structures, and hyphae of each of the following:
- Draw the conidia, conidia-bearing structures, and hyphae of three molds growing on the blood agar plates you exposed to
the environment. Do they resemble any of the fungi you observed previously?
- Draw the microscopic structures you have seen in each phase of a systemic fungus.