Serological Identification of Patient's Antibodies
The serology laboratory tests patients’ sera to detect specific antibodies. The results of such tests may provide a “serological diagnosis” of an infectious disease in which antibody was produced in specific response to the microbial antigens of the infecting microorganism. Demonstrating increasing quantities of antibody in the serum during the course of disease from its onset (when there may be little or no antibody) and acute stages through convalescence (when large amounts of antibody have been produced) constitutes evidence of current active infection and indicates the nature of the etiologic agent.The interaction of a patient’s antibody with a specific antigen may be demonstrated in one of several ways. Descriptive terms for antibodies refer to the type of visible reaction produced.
Agglutinins are antibodies that produce agglutination, a reaction that occurs when bacterial cells or other particles are visibly clumped by antibody combined with antigens on the cell surfaces.
Precipitins are antibodies that produce precipitation of soluble antigens (free in solution and unassociated with cells). When antibodies combine with such antigens, the large complexes that result simply precipitate out of solution in visible aggregates.
Antitoxins are antibodies produced in response to antigenic toxins. Since toxins are soluble antigens, in vitro interactions with antitoxins are seen as precipitation.
Opsonins are antibodies that coat the surfaces of microorganisms by combining with their surface antigens. This coating on the bacterial cell makes them highly susceptible to phagocytosis by white blood cells. (The word “opsonin” has a Greek root that means “to relish food.”)
Serological tests may be performed in vitro (in the test tube) or in vivo (in the body of an animal or human). In in vitro tests, such as those just described, quantitative methods are often employed. An in vivo test may employ experimental animals or cell cultures to demonstrate neutralization of an antigen by its antibody. When antibody combines with antigen in the test tube, the antigen is neutralized or inactivated. If it is a virus or other microorganism, it can no longer infect tissues or cell cultures. If it is a toxin combined with antitoxin, it is no longer toxic for tissues. If the antigen-antibody complex is injected into an animal, it will not cause disease or damage, whereas a nonimmune control animal injected with the antigen alone will display characteristic symptoms of disease, according to the nature of the antigen. Certain harmful antigens will also be ineffective when injected into animals if specific antibodies have been administered to the animal a short time before (passive immunity). Skin tests constitute another type of in vivo serological test. Depending on the nature of the antigen injected intradermally, a humoral or cellular (delayed hypersensitivity) immune response may be demonstrated. Patients immune to diphtheria experience no reaction at the site of injected diphtheria toxin (Schick test) because their circulating antitoxin neutralizes this antigen. Conversely, when persons who are (or have been) infected by tubercle bacilli are injected with a purified protein derivative of this microorganism, the response is a reddened area of induration at the injection site. This reaction results from vasodilation and infiltration of lymphocytes.
To quantitate antibody in serum, serial dilutions of the serum are made by setting up a row of test tubes, each containing the same measured volume of saline diluent. A measured quantity of serum is added to the first tube and mixed well. The dilution in this tube is noted (1:2, 1:4, 1:10, or whatever). A measured aliquot of this first dilution is then removed and placed in the second tube, containing measured saline. Material in the second tube is mixed, and an aliquot is removed and placed in the third tube. The procedure is repeated down the line of tubes, so that a graded series of serum dilutions is obtained. (This procedure is analogous to preparing antimicrobial dilutions as in Experiment 15.2.) The antigen is then added in a constant volume per tube. After allowing time (at the right temperature) for antigen-antibody combination to occur, the tubes are examined for visible evidence of such combination. The reciprocal of the last (highest) dilution of serum that produces a visible reaction is reported as the titer of the serum because it indicates the relative quantity of antibody present. If two sera are compared for reactivity with the same antigen, the one that can be diluted furthest and still show reactivity is said to have the higher titer, that is, the most antibody.
In serological diagnosis of infectious disease, it is almost always necessary to test two samples of the patient’s serum: one drawn soon after the onset of symptoms during the acute stage, and another taken 10 to 14 days later. The reason for this is that antibody production takes time to begin and to build up to detectable concentrations during the course of active infection. The first sample may show no antibody, or a low titer that could reflect either past infection or previous vaccination with the microbial antigen in question. If the second sample shows at least a fourfold or greater increase in titer as compared with the first, it is evident that current active infection has induced a rising production of antibody. Such laboratory information is of great value both in diagnosis and in evaluation of the immunologic status of the patient with respect to any antigen tested.
| Purpose | To demonstrate techniques for serological identification of patients’ antibodies |
| Materials | Demonstration material |
Results
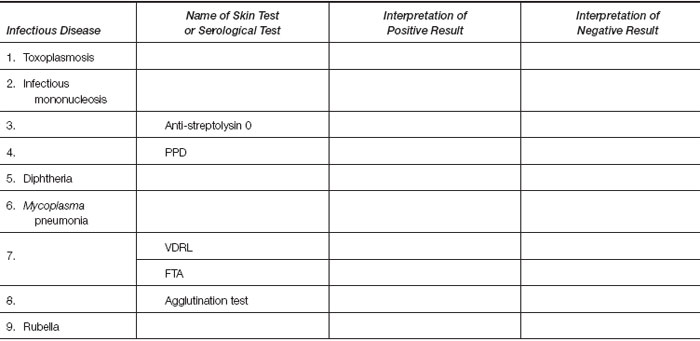
Review demonstration material and your reading assignments; then complete the following table.
 |




