Primary Media for Isolation of Microorganisms
As we have seen, many clinical specimens contain a mixed flora of microorganisms. When these specimens are set up for culture, if only one isolation plate were inoculated, a great deal of time would be spent in subculturing and sorting through the bacterial species that grow out. Instead, the microbiologist uses several types of primary media at once (i.e., a battery) to culture the specimen initially. In general, the primary battery has three basic purposes: (1) to culture all bacterial species present and see which, if any, predominates; (2) to differentiate species by certain characteristic responses to ingredients of the culture medium; and (3) to selectively encourage growth of those species of interest while suppressing the normal flora.The basic medium on which a majority of bacteria present in a clinical specimen will grow contains agar enriched with blood and other nutrients required by pathogens. The blood, which provides excellent enrichment, is obtained from animal sources, most often from sheep. The use of human blood (usually obtained from outdated collections in blood banks) in culture media is not recommended because it may contain substances such as antimicrobial agents, antibodies, and anticoagulants that are either inhibitory to the growth of fastidious microorganisms or interfere with characteristic reactions.
In addition to basic nutrients, differential media contain one or more components, such as a particular carbohydrate, that can be used by some microorganisms but not by others. If the microorganism uses the component during the incubation period, a change occurs in an indicator that is also included in the medium (see colorplate 16 and 17).
Selective media contain one or more components that suppress the growth of some microorganisms without seriously affecting the ability of others to grow. Such media may also contain ingredients for differentiating among the species that do survive.
When a battery of several culture media such as just described is streaked upon receipt of a clinical specimen, the first results indicate what types of bacteria are present, in general how many, and which did or did not use the differential carbohydrate. Also, the species of particular interest on the selective medium (if that species was present in the specimen) has been singled out and differentiated. Thus, the process of identification of isolated pathogens is already well under way after 24 hours of incubation of specimen cultures.
| Purpose | To observe the response of a mixed bacterial flora in a clinical specimen to a battery of primary isolation media |
| Materials | Nutrient agar plates Blood agar plates Eosin methylene blue agar plates (EMB) Mannitol salt agar plates (MSA) Simulated fecal suspension, containing Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus epidermidis Demonstration plates: Mannitol salt plate streaked with Staphylococcus aureus on one side (pure culture), Escherichia coli on the other (pure culture) Eosin methylene blue plate streaked with Staphylococcus aureus and Escherichia coli |
Table 16.1 summarizes the most commonly used enriched, selective, and differential media, indicating their purpose as primary media for the isolation of microorganisms. The table should be reviewed before performing the exercise.
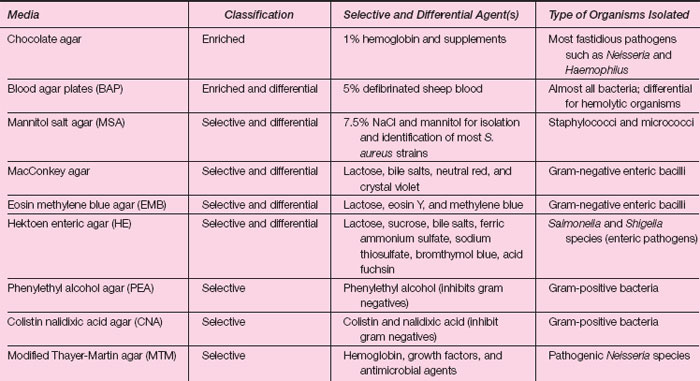 |
| Table 16.1 Culture Media for the Isolation of Pathogenic Bacteria from Clinical Specimens |
Procedures
- Inoculate the simulated fecal specimen on nutrient agar, blood agar, EMB, and MSA plates. Streak each plate for isolation of colonies. Incubate at 35°C.
- Make a Gram stain of the fecal suspension and examine it.
- Examine the demonstration plates (do not open them) and record your observations.
- Demonstration plates:
- Describe the appearance of S. aureus on
- Mannitol salt agar
- EMB agar
- Describe the appearance of E. coli on
- Mannitol salt agar
- EMB agar
- Describe the appearance of S. aureus on
- Simulated fecal specimen cultures.