Identification of Pure Cultures of Enterobacteriaceae from the Normal Intestinal Flora
| Purpose | To learn how enteric bacilli are identified biochemically |
| Materials | Slants of triple-sugar iron agar (TSI) SIM tubes MR-VP broths Slants of Simmons citrate agar Urea broths Slants of phenylalanine agar Lysine and ornithine decarboxylase broths Mineral oil in dropper bottle Sterile 1.0-ml pipettes Pipette bulb or other aspiration device Sterile empty test tubes Xylene Kovac’s reagent Methyl red indicator 5% alphanaphthol 40% sodium or potassium hydroxide 10% ferric chloride Nutrient agar slant cultures of Escherichia coli, Citrobacter koseri, Klebsiella pneumoniae, pigmented and nonpigmented Serratia marcescens, Enterobacter aerogenes, Proteus vulgaris, and Providencia stuartii |
Procedures
- Each student will be assigned two of the nutrient agar slant cultures. Inoculate each culture into the following media. TSI (using a straight wire inoculating needle, stab the butt of the tube and streak the slant; the closure should not be tight)
- SIM tubed agar (stab ¼ of the depth of the medium)
- MR-VP broth
- Simmons citrate agar slant
- Urea broth
- Phenylalanine agar slant
- Lysine decarboxylase broth
- Ornithine decarboxylase broth
- Carefully overlay the surfaces of the lysine and ornithine broths with ½ inch of mineral oil.
- Incubate all subcultures at 35°C for 24 hours.
- Before returning to class, read the following descriptions of the biochemical reactions to be observed and instructions for performing them.
Biochemical Reactions and Principles
- TSI:
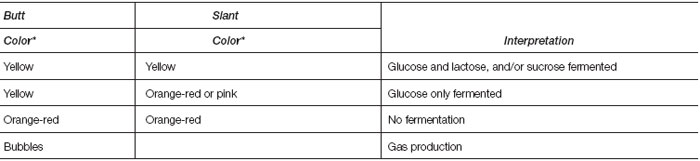
TSI contains glucose, lactose, and sucrose as well as a pH-sensitive color indicator. It also contains an iron ingredient for detecting hydrogen sulfide production, which blackens the medium if it occurs (compare with H2S detection in SIM medium). Kligler’s Iron Agar is similar but sucrose has been omitted (see colorplate 19).
Fermentation of the sugars by the test organism is interpreted by the color changes in the butt and the slant of the medium. - B. IMViC Reactions:
The term IMViC is a mnemonic for four reactions: the letter I stands for the indole test, M for the methyl red test, V for the Voges-Proskauer reaction (with a small i added to make a pronounceable word), and C for citrate.
The indole test for tryptophan utilization was described in Experiment 17.3. Perform it in the same way here, using xylene and Kovac’s reagent added to SIM cultures.
Methyl red is an acid-sensitive dye that is yellow at a pH above 4.5 and red at a pH below 4.5. When the dye is added to a culture of organisms growing in glucose broth, its color indicates whether the glucose has been broken down completely to highly acidic end products with a pH below 4.5 (methyl red positive, red), or only partially to less acidic end products with a pH above 4.5 (methyl red negative, yellow).
The Voges-Proskauer test can be performed on the same glucose broth culture used for the methyl red test (MR-VP broth). One of the glucose fermentation end products produced by some organisms is acetylmethylcarbinol. The VP reagents (alphanaphthol and potassium hydroxide solutions) oxidize this compound to diacetyl, which in turn reacts with a substance in the broth to form a new compound having a pink to red color. VP-positive organisms are those reacting in the test to give this pink color change.
To perform the MR and VP tests, first withdraw 1.0 ml of the MR-VP broth culture, place this in an empty sterile tube, and set the tube aside for the VP test. Discard the pipette in disinfectant.
Do a methyl red test by adding 5 drops of methyl red indicator to 5.0 ml of MR-VP broth culture. Observe and record the color of the dye.
Perform a VP test by adding 0.6 ml of alphanaphthol and 0.2 ml of KOH solutions to 1.0 ml of MR-VP broth culture. Shake the tube well and allow it to stand for 10 to 20 minutes. Observe and record the color.
Citrate can serve some organisms as a sole source of carbon for their metabolic processes, but others require organic carbon sources. The citrate agar used in this test contains bromthymol blue, a dye indicator that turns from green to deep blue in color when bacterial growth occurs. If no growth occurs, the medium remains green in color and the test is negative.
-
Motility and H2S Production:
These properties are observed in SIM cultures, as described in Experiment 17.3. -
Urease Production:
The test for urease was described in Experiment 18.1. Read and record the results of your cultures tested in urea broth. -
Phenylalanine Deaminase (PD):
The test for production of this enzyme was described in Experiment 18.5. Perform it in the same way, adding ferric chloride solution to your cultures on phenylalanine agar medium. -
Lysine (LD) and Ornithine (OD) Decarboxylases:
Lysine and ornithine are amino acids that can be broken down by decarboxylase enzymes possessed by some bacteria. During this process, the carboxyl (COOH) group on the amino acid molecule is removed, leaving alkaline end products that change the color of the pH indicator. In the broth test you use, a positive test is a deep purple color; a negative test is yellow. The reactions work best when air is excluded from the medium; therefore, the broths are layered with mineral oil after inoculation and before incubation.
 |
Results
Record results for your cultures in the following table. Obtain results for other cultures by observing those assigned to fellow students.
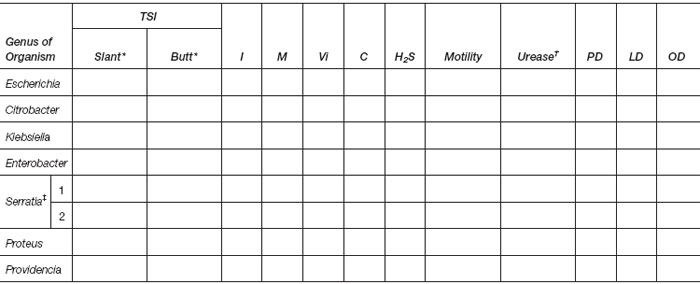 |
†If positive, specify time.
‡1 = pigmented strain; 2 = nonpigmented strain.




