13C-NMR spectra
The 13C nucleus has I = ½ like 1H, and the 13C-NMR spectrum of a compound can be observed using a different radio frequency range (in the same magnetic field) to that for 1H. The 13C spectrum will give peaks for each different type of carbon atom in a molecule but the properties of the 13C nucleus give some important and useful differences in the spectrum obtained:- The natural abundance of 13C is only 1.1% compared with 98.9% for 12C - in any molecule no two adjacent atoms are likely to be 13C and therefore coupling between 13C nuclei will not be seen, giving a very simple spectrum.
- In a sample of a compound, which contains many molecules, the 13C isotope is randomly distributed and all the different carbon atoms in a sample of a compound will be seen in the 13C-NMR spectrum.
- The sensitivity of the 13C nucleus is low and this, together with its low natural abundance, means that FT -NMR is the only practical system to produce a spectrum by accumulation of spectra by repetitions. Larger sample size in bigger NMR tubes also assist in solving the sensitivity/ abundance problem.
- The chemical shift range for 13C is greater (δC = 0-250 ppm) than for 1H (δH = 0-15 ppm) giving greater spectral dispersion, i.e. the peaks for carbons with very slight differences in chemical shifts are separated and do not overlap.
- 13C nuclei will couple with the 1H nuclei to which they are directly bonded, e.g. CH3 will appear as a quartet, CH2 as a triplet, CH as a doublet, but C with no hydrogen atoms attached will appear as a singlet. This introduction of complexity in the 13C-NMR spectrum is removed by broadband decoupling.
- The peak areas of the different carbon atoms are not related to the number of carbon atoms having the same chemical shift, as in the case for 1H-NMR spectra.
Interpreting 13C-NMR spectra
Normally you will be given two 13C-NMR spectra (Fig. 29.11). The upper spectrum, which is more complex (more lines) is called the off-resonance decoupled spectrum and shows the 13C–1H coupling to enable you to determine which carbon signals are CH3, CH2, CH and C. Then overlapping of peaks may make the identification of different carbon atoms difficult. The lower spectrum is a broadband decoupled spectrum in which the molecule is irradiated with a second radio frequency range for the protons in the molecule and effectively removes all the 13C–1Hcouplings from the spectrum. The resulting simplicity of the spectrum makes identification of the different types of carbon in the molecule relatively easy.
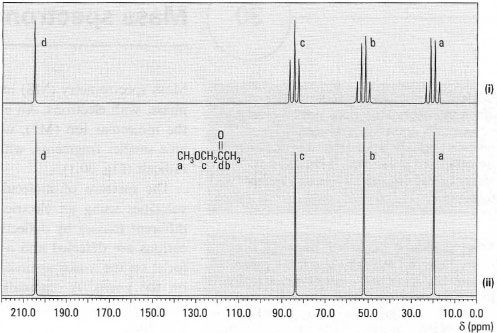 |
| Fig. 29.11 13C–NMR spectra of 1-methoxypropanone: (i) off-resonance decoupled; (ii) broadband decoupled. |
The chemical shifts of 13C atoms (δC) vary in the same manner as those of protons (Fig. 29.12):
- δC moves downfield as the hybridization of the carbon atom changes from sp3 (0-50 ppm) to sp (75-105 ppm) to sp2(100-140 ppm);
- for sp3 hybridized carbon: δC moves further downfield with the change from CH3 to CH2 to CH to C;
- for sp2 hybridized carbon: aromatic carbons occur further downfield (δC = 115-145 ppm) than alkene carbon atoms (δC = 100-140 ppm);
- bonding more electronegative atoms to carbon deshields the carbon atom and moves the peaks downfield, e.g CH3C (δC ~ 6 ppm) and CH3O (δC ~ 55 ppm), C=C (δC ~ 123 ppm) and C=O (δC ~ 205 ppm).
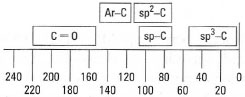 |
| Fig. 29.12 Approximate chemical shift positions in 13C-NMR. |




